#52 Flux materials in ceramics: definitions & mechanisms of action
- Flux materials: what are they and how they are used
- Types of flux materials
- What happens on a molecular scale
- Flux materials in ceramic production
- Digital glues: fluxes & areas of influcence
1. Flux materials: what are they and how they are used
A flux is a solid substance that plays a key role in lowering the melting point of materials, helping them fuse together at lower temperatures.
Essentially, it speeds up the sintering process by making the glassy phase melt sooner and more easily.
SINTERING is a high-temperature process that transforms powdered material—made up of tiny individual particles—into a solid, unified structure.
It’s commonly used to create materials with specific properties that wouldn’t be achievable through conventional manufacturing methods.
Essentially, sintering densifies the powder compact, significantly reducing internal porosity.
This technique, also applied to metals and polymers, allows the material to solidify at temperatures lower than its melting point. For simple shapes, it can even eliminate the need for additional machining or finishing steps.
In the ceramics industry, fluxes are widely used for a variety of purposes, depending on the specific application—whether in body formulations, glazes, or specialized products like digital glues.
In the case of digital glues, fluxes are primarily valued for their ability to interact with glass frit. During the firing process, this interaction promotes the melting of the frit, allowing it to bond more effectively to the ceramic substrate through the glue. The result of this interaction is typically a smoother, more uniform surface with improved flatness and enhanced visual quality. In short, adding a flux to the digital glue improves how the frit behaves during firing, leading to better aesthetic and technical results.
From a chemical standpoint, discussing ceramic fluxes largely means referring to raw materials or frits that contain alkali metals like sodium and potassium. These elements are typically used in controlled proportions, often combined with others such as boron, calcium, magnesium, or zinc.
Some of these substances have a lower melting point than silica—which, as is well known, is the primary glass-forming component in glazes and frits. In other cases, flux materials are valued for their ability to form eutectic mixtures with silica. These are special blends that melt at a lower temperature than any of the individual components alone—hence the term “easy to melt.” Even when the oxides involved have higher melting points than silica on their own, combining them with silica can result in mixtures that melt more readily than the pure substances would individually.
To give a practical example, let’s look at the simplest case: Silica (SiO₂) is the main component in glass and glazes, and one of the most important cross-liking forming oxides. However, it has a very high melting point—around 1713°C—which makes it difficult and costly to use in its pure form.
On the other hand, materials like sodium oxide or potassium oxide melt at much lower temperatures.
When combined with silica, they alter its structure, effectively lowering the temperature at which it begins to melt.
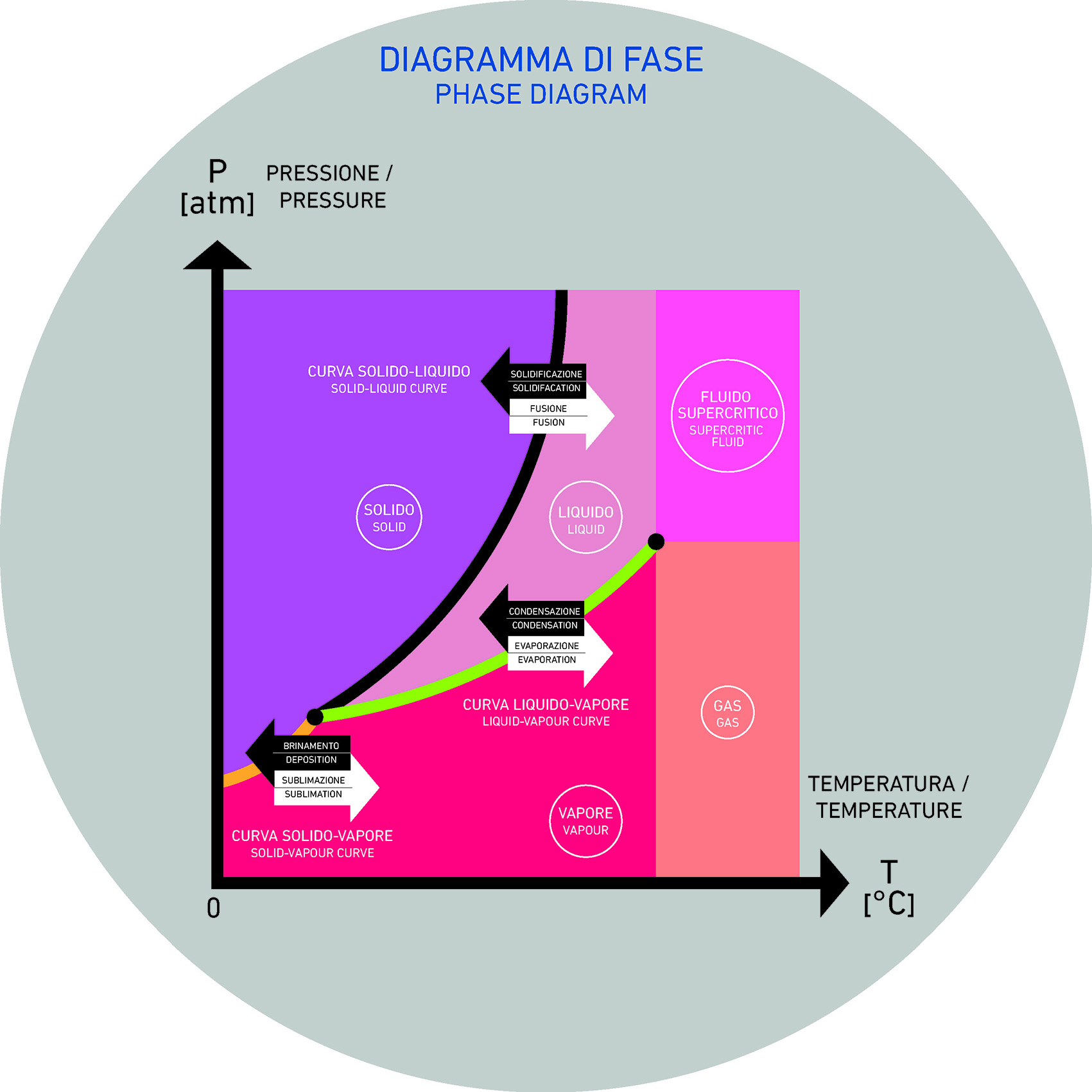
One important aspect to keep in mind is that when these materials are mixed with silica, they help lower the viscosity. From a practical standpoint, assuming all other conditions are equal, this results in a clear processing advantage. Ultimately, the role of a flux is to reduce the compound’s melting temperature, producing a more fluid melt than would be possible using pure silica alone.
To complete the picture, it’s worth noting that ceramic glazes don’t just rely on fluxes and glass-forming components. A third key ingredient is a stabilizer, most commonly aluminum oxide (Al₂O₃), which is typically introduced via feldspars and clays. Although aluminum oxide is not a flux—in fact, it’s a refractory material—it plays an essential role in controlling the melt’s viscosity and promoting vitrification during cooling.
Aluminum is the second most abundant element in ceramic glazes, after silicon, and it contributes significantly to the structural strength and stability of the final product.
2. Types of flux materials
There are several types of flux materials, each capable of providing specific properties to the system in which it’s used. Depending on the characteristics you want to achieve in the glass or glaze, it’s essential to carefully select the appropriate flux in advance—and to test it according to the desired result. Regardless of the specific goal, one constant is that fluxes change the behavior of the silica they’re mixed with—including its mechanical properties. Depending on the application, various performance factors may come into play, such as scratch resistance, water solubility, chemical durability against acids and alkalis, and shrinkage control*, along with many other functional attributes.
When referring to materials with fluxing properties, we’re generally talking about oxides.
These substances are rarely used in their pure form; instead, they are typically introduced into glazes through raw materials or frits. This is because each flux imparts its own distinct properties to the glaze.
Sodium oxide and potassium oxide, for instance, are considered primary fluxes. However, it’s important to note that these oxides can also impact the chemical resistance of the glaze.
For this reason, they are often incorporated via frits or feldspars, which help modulate and balance their effects.
Calcium oxide is a highly effective flux that helps stabilize the chemical and mechanical properties of glass. However, careful dosing is essential, as excessive amounts can lead to increased surface opacity. It is most introduced into formulations in the form of calcium carbonate.
Magnesium oxide is considered a secondary flux, often used alongside others to help control the glaze’s thermal expansion coefficients. Typical raw materials that supply it include talc and dolomite.
Boron, usually added through boron frits, plays a versatile role—acting as a flux, a stabilizer, and a vitrifying agent all at once.
Zinc oxide is also classified as a secondary flux and is typically used to promote the formation of matte or opaque surfaces.
All these materials can be used not only in glaze or frit formulations but also in digital glues.
Here too, the type of flux and its percentage vary depending on the intended function and performance goals. In short, the initial formulation choices have a direct impact on the final properties of the glass. There is a wide range of glass types, just as there is a wide variety of fluxes available, meaning that combinations and dosages are highly variable and cannot be standardized.
SHRINKAGE BEHAVIOR: In general, when glass is heated, it expands; conversely, as it cools, it contracts.
This shrinkage or expansion is governed by the material’s thermal expansion coefficient, which is directly influenced by its chemical composition. A simple comparison illustrates this: think of the different behaviors of Pyrex glass, window glass, and a smartphone screen. Although all are types of glass, each exhibit distinct mechanical properties.
Pyrex (a registered trademark) is a special type of glass with a very low thermal expansion coefficient, making it highly resistant to thermal shock. That’s why it’s commonly used in laboratory equipment, industrial applications, and cookware.
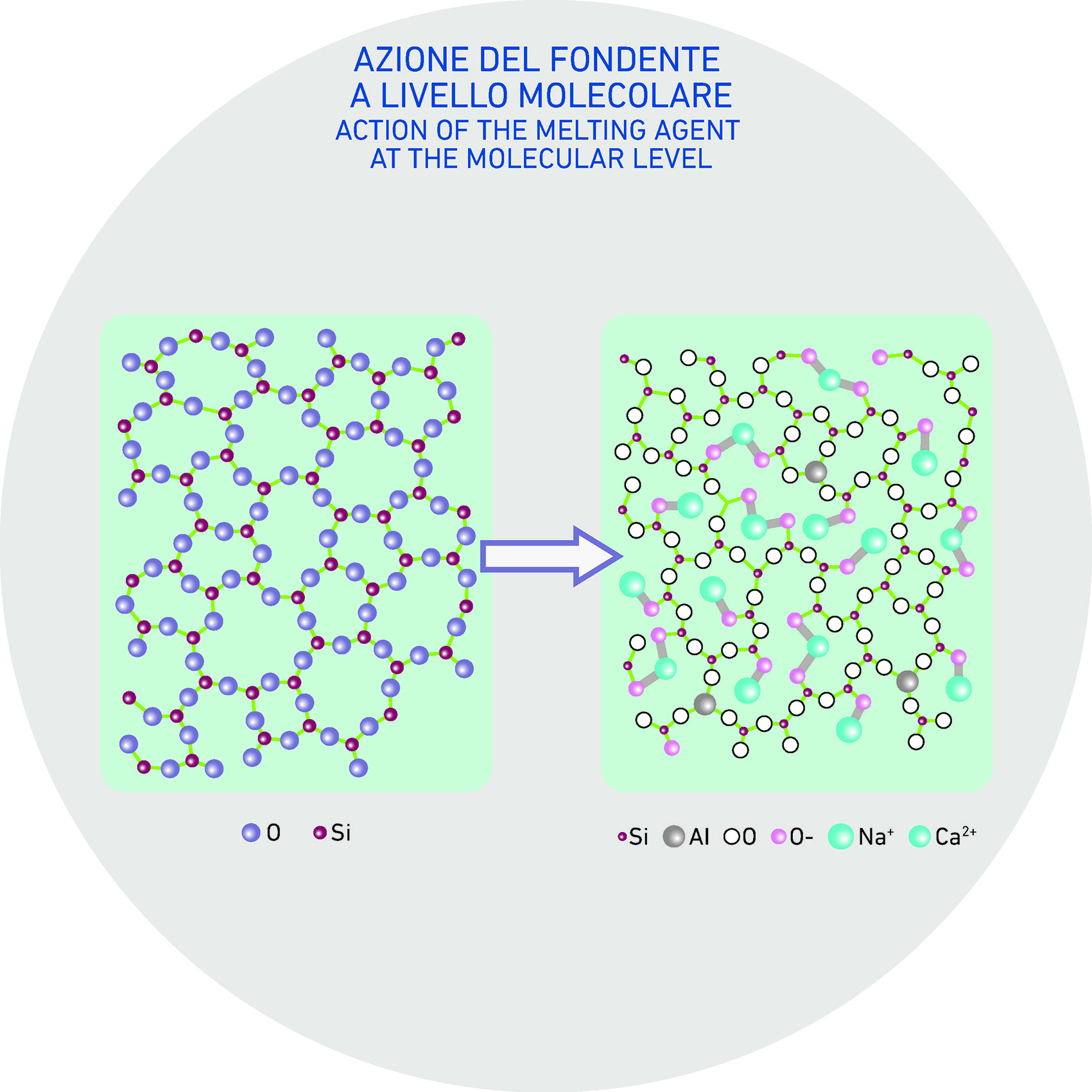
3. What happens on a molecular scale
The action of fluxes—especially oxides of alkali and alkaline earth metals—facilitates the breakdown and modification of silica’s three-dimensional structure. This effect is primarily due to the presence of monovalent cations (such as sodium and potassium) and divalent cations (such as calcium) found in fluxing agents. These cations integrate into the silica, disrupting its rigid lattice.
This structural modification leads to several effects, most notably a reduction in viscosity and the well-known lowering of the melting temperature.
4. Flux materials in ceramic production
The use of flux materials in the ceramic production cycle—particularly in digital glues—can, in some cases, bring significant advantages for manufacturers, who are often supportive of their presence for this very reason.
The enhanced and faster melting behavior of frits or glazes can in fact be seen as an energy-related benefit: shorter firing times translate directly into cost savings and improved production efficiency.
It’s important to note, however, that the action of fluxes is not inherently beneficial across all areas of ceramic production.
In certain sectors—such as refractory materials—fluxes, particularly sodium and potassium (which are among the most reactive), are generally avoided or kept to a minimum.
This is because one of the key goals in refractory manufacturing is to maximize the melting point of the material, given its intended use in high-temperature applications. In this context, the presence of fluxing agents would compromise the product’s performance rather than enhance it.
MATTE FLUXES AND GLOSSY FLUXES
Can we talk about macro-categories of fluxes?
It’s a fair question, even if not easy to answer definitively. While the terminology is far from scientific, it is common—at least informally—to distinguish between matte fluxes and glossy fluxes.
But what determines the final appearance of a tile’s surface?
Let’s start with a basic principle: silica-based glass is naturally glossy and transparent. To create a matte effect, its composition must be altered—and there are several ways to achieve this. One common approach is to add substances that, during the firing process, modify the glass composition and promote non-uniform crystallization during cooling. Alternatively, a partially insoluble raw material may be introduced into the silica matrix to achieve a similar effect.
In both cases, the result of firing is the formation of multiple solid phases within the glass—each with different chemical compositions. At the boundaries between these phases, light is no longer transmitted in a straight path but rather scattered in different directions. This light diffusion is what gives the surface its matte appearance. Conversely, a glossy glass does not exhibit such phase differences; it remains a homogeneous phase throughout, allowing light to pass through smoothly and evenly.
5. Digital glues: fluxes & areas of influcence
Given all the above, it’s important to emphasize that the effectiveness and melting behavior of a flux can vary significantly from one customer to another. That’s because the glue itself is not an independent variable—it must always be considered in relation to both the frit and the firing cycle.
The optimal result stems from the balance between these three elements. For this reason, it’s not possible to make absolute claims about the benefits or limitations of any given glue. A particular formulation may offer clear advantages in one context yet prove completely unsuitable in a different production setup. It always depends on the perspective and the specific combination of materials and conditions at play.
While the presence of a flux doesn’t significantly affect the adhesive strength of the glue, it can influence several key performance areas—some of which we’ll explore below.
Depending on the type of flux used, interactions with color inks can vary in intensity—sometimes enhancing certain tones over others. In general, fluxes always have some impact on color development: in some cases, the effect is subtle; in others, it’s more pronounced.
Whether this interaction proves beneficial or counterproductive depends entirely on the intended final product.
One of the main outcomes of using flux is a more even and consistent distribution of the frit.
This is due to the higher degree of melting, which increases the fluidity of the glass, allowing it to spread more uniformly across the ceramic surface.
Achieving proper frit fusion—with good transparency and minimal defects—at lower temperatures than would be possible without fluxes can ideally lead to reduced energy consumption during firing.
This is especially true when the glaze-glue-frit system, combined with the appropriate firing cycle, results in both technically sound and aesthetically pleasing outcomes.
That said, a flux is still a raw material, and like any raw material, it can introduce undesirable side effects. This is why it’s crucial to carefully tune the formulation and thoroughly evaluate the interactions between the glaze, glue, frit, and firing conditions—to avoid potential issues later in the production process.
For example, a flux within the glue might act too aggressively on the glass grit, causing the melted glass to become overly fluid, potentially leading to boiling or bubbling defects. In such a scenario, corrective action might be needed—whether by adjusting the glue formulation, the frit composition, or (though less likely) the firing schedule—to reestablish the right balance among the system’s components.
In most cases, these are process-related issues rather than product-related ones.
There are instances, for example, where someone might argue that the presence of a flux in the glue causes the well-known black core effect inside the tile body. However, the real issue often lies not in the glue itself, but in how the production process is configured. As mentioned before, that same glue might perform perfectly well under different conditions.
One critical concern—or at least an aspect that requires special attention—when using fluxes in glues is the potential for particle sedimentation. If not properly managed, flux-containing formulations may experience a settling of solids over time. Although glues are typically formulated to minimize this phenomenon, it’s still essential to ensure proper suspension management. For example, using automated storage systems that continuously stir the material and feed the digital printer with a fully homogenized product can help prevent inconsistencies.
Even if minimal, some solid content will inevitably settle. If the glue is not re-homogenized before use, the result may be inconsistent application: residual solids left at the bottom of the container mean the printer may apply glue with a lower solid content than in previous batches—compromising performance. Fortunately, as you might expect, these types of issues are easily solvable with the right process controls.

