#39 Cratering phenomena: what are they, how they are formed, & how to handle them
- Identikit
- Glazes & grits suspensions (high & low weight)
- Origins & causes:
- Impurities in the ceramic raw materials
- Improper use defoamers
- Improper application parameters
- Contamination of the suspension
- Conclusions
1. Identikit
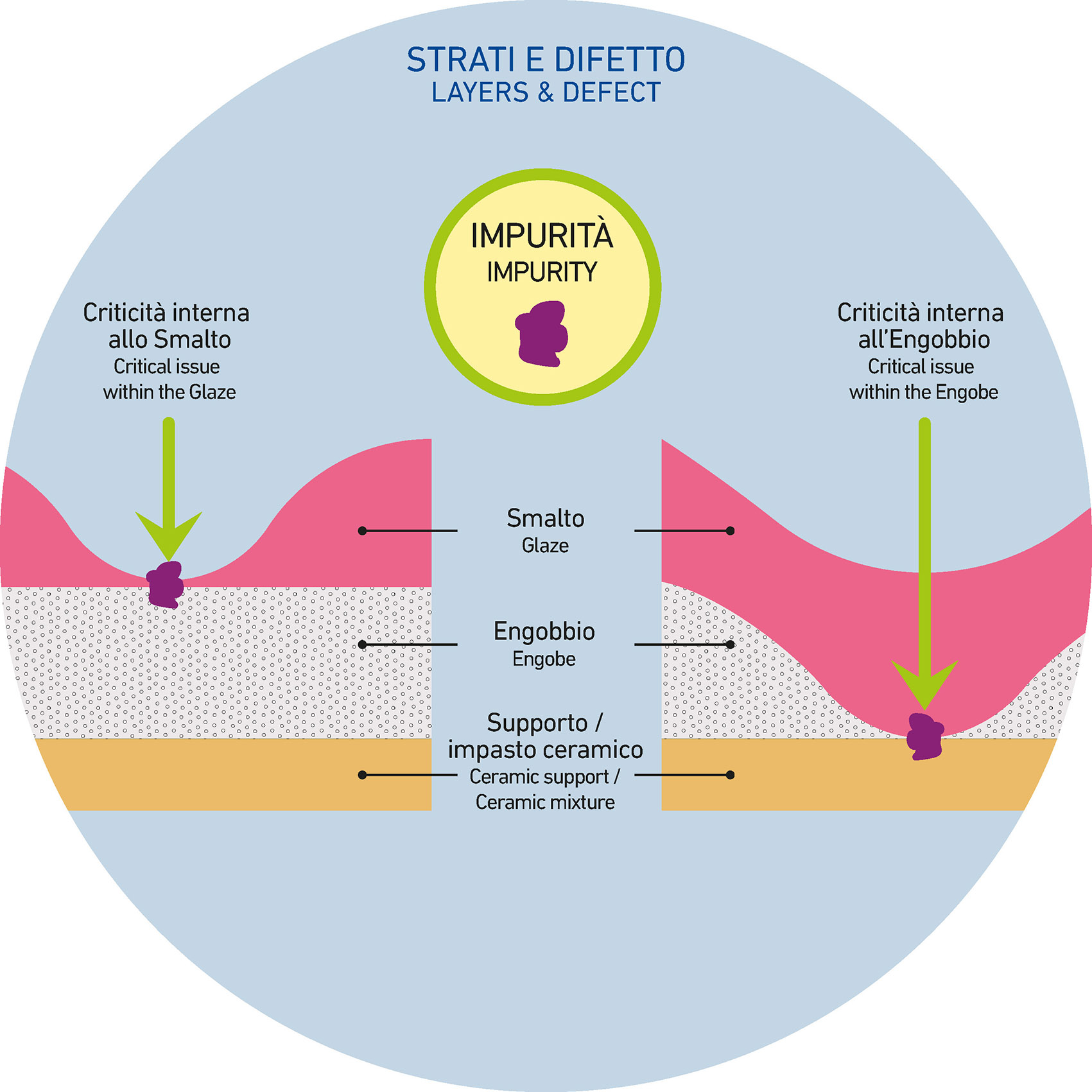
Cratering phenomena are defects that appears as microscopic craters on the surface of the ceramic support where an aqueous suspension has been applied and is still in the drying phase. This issue results from an imperfect application due to one or more depressions, causing a lack of perfect flatness in the layer of suspension just applied.
The depth of the resulting “crater” may vary, and the defect can affect the top layer (such as the glaze) or other previously applied underlying layers, like engobe or smaltobbio.
What are the differences between this defect and a pinhole?
The difference is very little but important, as both are defects that can compromise the flatness of the surface obtained after firing. Generally speaking, it can be said that a pinhole is a defect that largely becomes visible only after firing, developing in response to the temperatures and processes within the kiln. The “crater”, on the other hand, is an issue visible already during the application phase, thus appearing in the stages of the ceramic production process prior to firing.
2. Glazes & grit suspensions: high and low weight applications
The presence of micro-craters usually affects glaze and engobe suspensions, especially when applied at low weight (low solid content). One could even say that pinholes rarely appear in conjunction with wet grit applications, even in case of a low solid content. Why?
High-weight grit suspensions, so called due to the significant amount of solid material they contain, are marked by particles size (often larger than that of glaze suspensions) that requires the use of specific categories of suspending agents. These additives help to get the proper rheological parameters while ensuring the suspension of the solid component, which has a notable size and mass. Both the rheology of the suspension and the grit grain size can inhibit or at least limit the occurrence of the problem.
Let’s simplify and start with two statements:
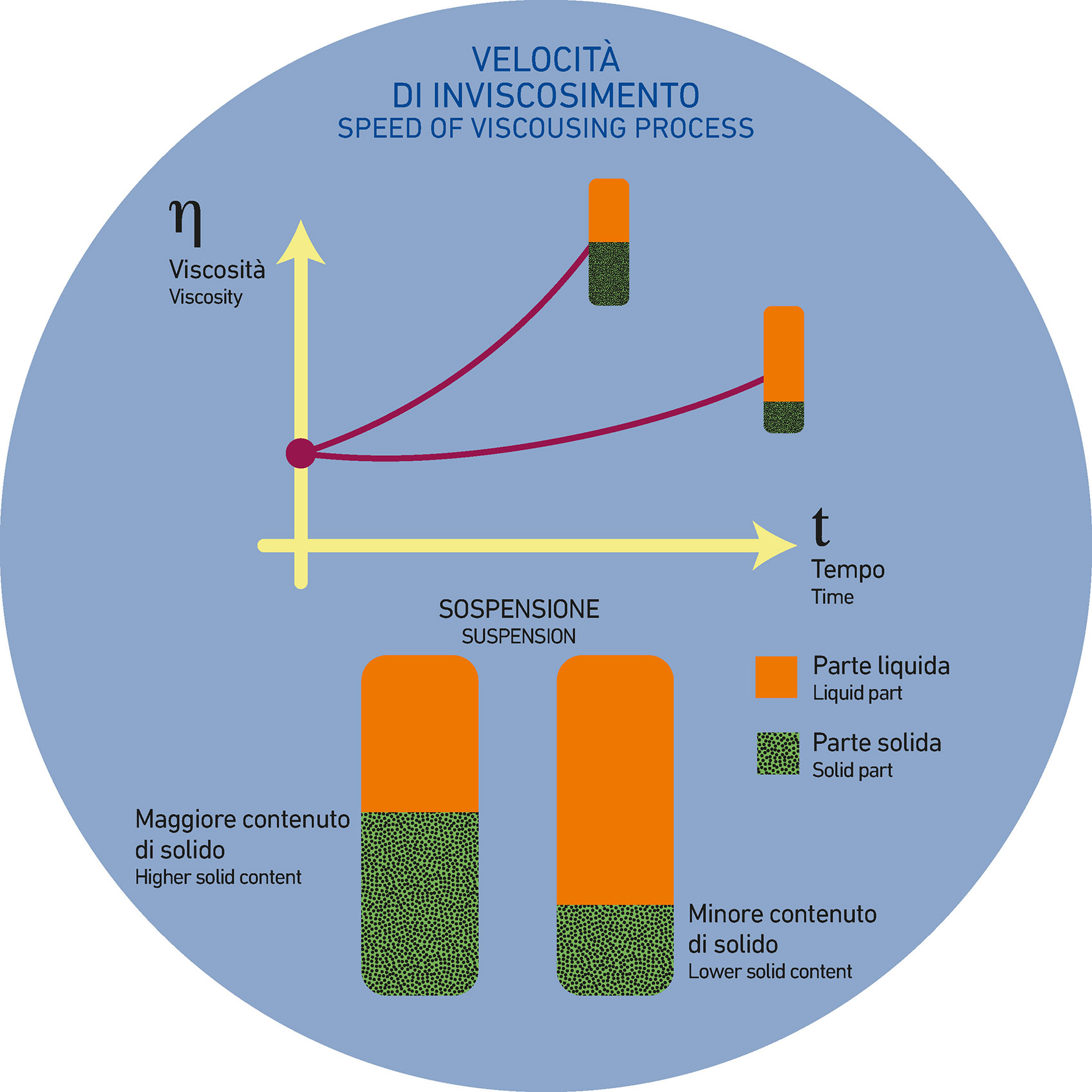
- High weight / high density = more solid and less water
- Low weight / low density = less solid and more water
From an application standpoint, the different features of these two types of suspensions imply the following: the suspension is spread onto the ceramic support and, as usual, begins to lose its water content and then dry, due to evaporation and drainage processes. This phenomenon naturally leads to a progressive increase of the viscosity of the suspension. Up to this point, both suspensions behave similarly.
However, the rate at which viscosity increases will be surely different: the high-weight suspension, having less water to release, will take less time compared to the low-weight suspension, which will require more time to reach the same (hypothetical) level of viscosity.
The longer duration of the process, if the system is not well-calibrated, can somehow influence the PROBABILITY of the defect occurring.
Drying times for grit suspensions are generally shorter than those for glaze suspensions, given that the particle size of the grit is significantly larger. The size influences drainage times because a larger particle size usually corresponds to faster drainage. This aspect, due to the greater speed, helps reduce the likelihood of the defect appearing.
3. Origins & Causes
The appearance of a pinhole, like any other defect affecting the ceramic body, is usually due to a combination of factors that may also interact with each other, making the list of possible causes quite extensive. To simplify, we outline below the most significant and impactful scenarios.
- Impurities in the ceramic raw materials
- Improper use of defoaming additives
- Incorrect application parameters: excessively long drying times and density/viscosity values that are too low for the parameters of the production line
- Contamination of the suspension
- IMPURITIES IN THE CERAMIC RAW MATERIALS
As we known, glaze suspensions contain not only frits but, in smaller proportions, also the raw materials that make up ceramic mixtures: clays, feldspars, etc.
Since they come from natural caves, these materials can sometimes contain organic, carbon-based impurities. In this regard, clays are the main responsible, and even though their percentage in the suspension is low, they can contaminate the mixture if such impurities are present.
High-carbon residues, when ground, often prove incompatible with the system and struggle to be wetted by water. This phenomenon results, during application, in the breaking of the glaze film due to the impurity that somehow “repels” the glaze suspension that try to cover it. This action is precisely what creates and leads to cratering phenomena.
How can we address and eliminate the problem?
It is generally advisable to use an appropriate surfactant specifically designed for the suspension to which it will be added. The surfactant, in addition to providing a levelling effect, makes the impure components (carbon-based impurities) compatible with the system.
How does it work?
By positioning itself at the interface between the “impure” components and the liquid part of the suspension, the surfactant molecules—typically composed of a hydrophilic head and a hydrophobic tail—surround the solid parts of the incompatible elements, encapsulating and making them compatible. This action promotes proper application, preventing the breaking of the film of the glaze, engobe, or smaltobbio.
Usually, selecting the correct surfactant allows the process to continue without further adjustments to production parameters.
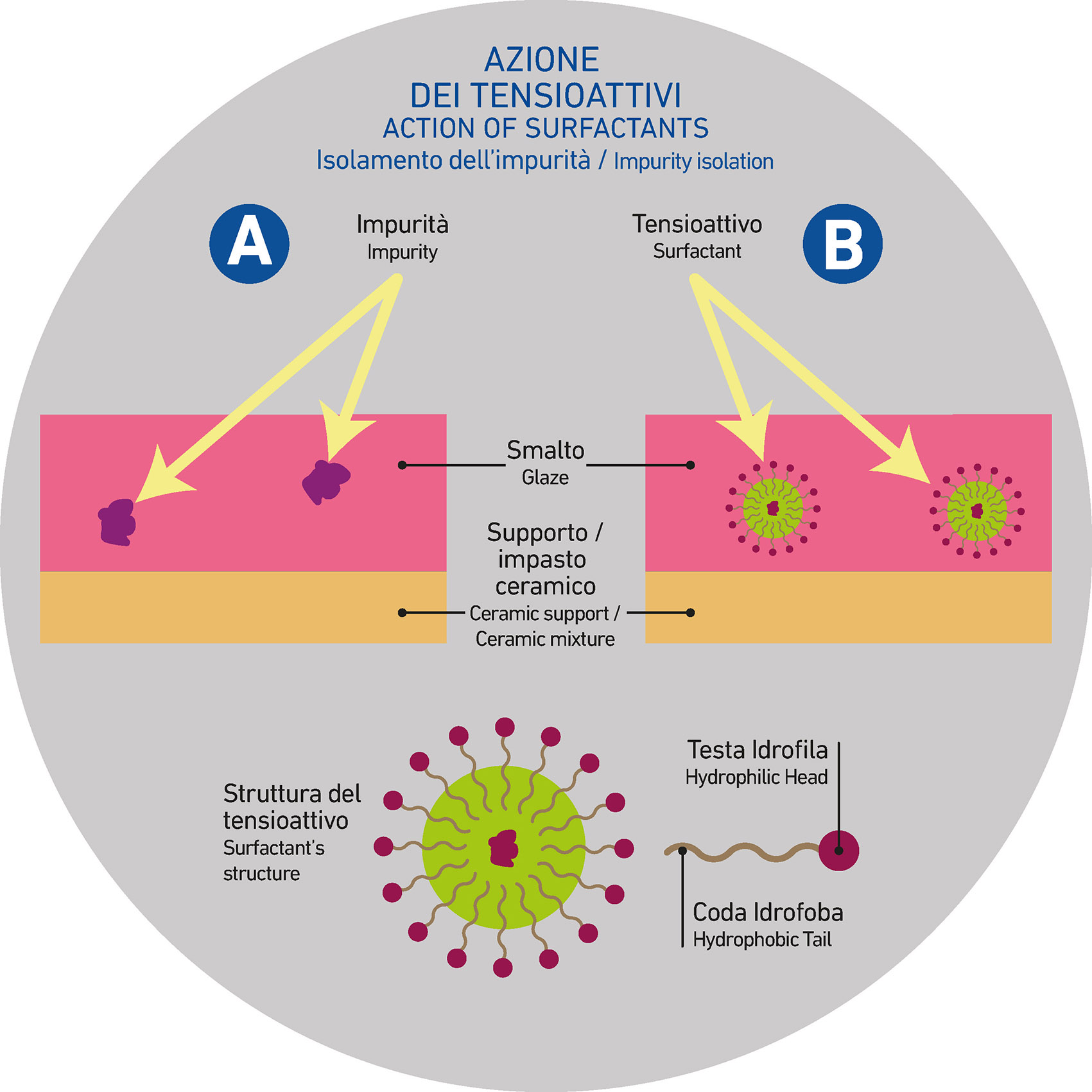
Is the choice of the type of surfactant irrelevant?
As previously mentioned, surfactants are not all the same and can be marked by significantly different chemical and physical properties. Therefore, there are no guarantees about their universal effectiveness. Some surfactants, when added to certain suspensions, may be less strong or ineffective—not because they are wrong, but because they are not suitable for that particular system.
Sometimes, they may even cause additional problems. For example, some surfactants are developed and designed to work within high-density systems (such as for bell application systems) to achieve excellent levelling. The same surfactants, when used in airless application systems, which involve lower density suspensions, may not be as suitable because the liquid-solid ratios are not appropriate for that type of addition.
The surfactant must therefore be selected based on rheological values, the type of application (vela, bell, airless, valves), working density, and, more generally, other relevant parameters.
- IMPROPER USE OF DEFOAMERS
Defoamers are chemical agents effective in preventing or eliminating air bubbles that can sometimes form and trapped within the suspension and/or on its surface. These bubbles may emerge inside the stirring tank and can even persist after application.
In general, defoamers, being partially water insoluble, have the ability to destabilize the foaming system. By spreading rapidly, they act on the equilibrium formed at the liquid-air interface (which is the basis of foam bubbles), breaking the lamellae and causing the bubble to burst.
Defoamers are usually effective because they are partially incompatible with the suspension. This partial incompatibility is due to their polarity or, more precisely, their hydrophilic-lipophilic balance, which measures their degree of hydrophilicity or lipophilicity (i.e., their affinity for fats or water).
So, why do we talk about improper use?
Generally speaking, when applied to less plastic glazes (with low clay content) or hard glazes (such as glazes based exclusively on frits), defoamers tend to remain micro-dispersed in water, sometimes causing defects. Among these, pinholes or cratering phenomena are quite common. The use of defoamers to counteract foam promoted by surfactants may partially or fully solve the problem, but the resulting balance can sometimes be unstable.
As with surfactants, there are different types of defoamers. To contain or prevent the appearance of cratering phenomena they may cause, several approaches can be taken. The most immediate and popular is to review and replace the defoamer with a type that better combines with the surfactant in the suspension, if present. [Given the vast number of cases, it does not make sense to provide an example here.]
Additionally, it may be helpful to adjust the dosage, either reducing or increasing the amount based on the rheological properties of the mixture. Another possible approach is to review the surfactant: since defoamers and surfactants work in combination, adjustments can be made to either or both components.
- IMPROPER APPLICATION PARAMETERS
Among the numerous parameters that could be considered, we focus here on two in particular, which are interrelated: excessively long drying times and too low density/viscosity values in relation to the parameters of the production line.
This discussion is somewhat linked to what was presented in paragraph two.
During the drying of the suspension that has been discharged onto the substrate, various phenomena can occur. If the drying/draining process is excessively long (we are talking about a few seconds: from a few seconds to several tens of seconds), the suspension, instead of solidifying quickly, remains fluid for too long. This time frame is enough to facilitate the appearance of defects. For instance, if a carbon impurity or a drop of defoamer is present, the likelihood of the suspension film breaking would be significantly higher.
If time, therefore, is a significant parameter, it’s clear how important it is, in such cases, to act by reducing and re-calibrating the drying times. What are the most effective actions?
A) Increase the application density by reducing the amount of water in the solution, thereby shortening evaporation/drainage times and consequently lowering the risk of pinholes. To reduce the water content and increase the density without drastically modifying the parameters of the production line, one could use strong dispersants that facilitate proper processing of the mixture.
In other cases, a binder may be used, which does not excessively raise the system’s viscosity values and thus allows for operation with a lower water content. The viscosity of the selected binder (or glue) indeed affects the system and can contribute to increasing density values.
In extreme cases, when all measures seem ineffective, the density increasement may require new fluidization study within the labs. It is important to remember that the decision to work at high or low densities always depends on the tareget and on the line parameters. One is not necessarily better than the other.
B) Review/adapt the glazing line, for example, by positioning the spray cabin (or the application machine) further upstream on the line, where the ceramic body temperature is higher, thereby accelerating evaporation/drying times. From this point of view, if there are two cabins available, it would clearly be more appropriate to use the one closer to the dryer.
- CONTAMINATION OF THE SUSPENSION
Sometimes the suspension can be contaminated during the application phase along the glazing line.
This can happen particularly when, after finishing one application, production continues with a different product that requires the use of a new suspension (characterized by different parameters and additives). If great care is not taken to clean the application machine before starting the new production (whether it is an airless spray cabin, a vela, a bell, etc.), there is a risk of contaminating the new suspension.
What happens?
The new suspension has different characteristics and contains different chemicals, yet it is affected by the organic (and other) residues left on the machine by the previously used suspension. Some defoamers, for example, can deposit on the walls of the spray cabin, inside the tank, and within the pipes. If thorough cleaning is not performed at shift or production changes, the greasy and oily residues remain attached to the metal and can interact with the new suspension. However, since these residues are incompatible and there is no appropriate surfactant to keep them in emulsion, they may lead to the formation of cratering phenomena.
In this situation, the best solution to solve the problem is to carry out a thorough cleaning of the machine using appropriate cleaners that can solubilize the oily parts of the defoamer. Another good practice is to limit or eliminate the use of those categories of defoamers that tend to cause such issues, thereby reducing dependency from one production to the next.
4. Conclusions
From this brief explanation, we should learn an important lesson.
Achieving excellent application results and inhibiting (or preventing) critical issues that can compromise both the product and the process always means finding the right balance among the elements involved. Adjusting one parameter may worsen another and vice versa. The challenge is to define which levers to adjust in order to achieve a customized and well-balanced outcome.
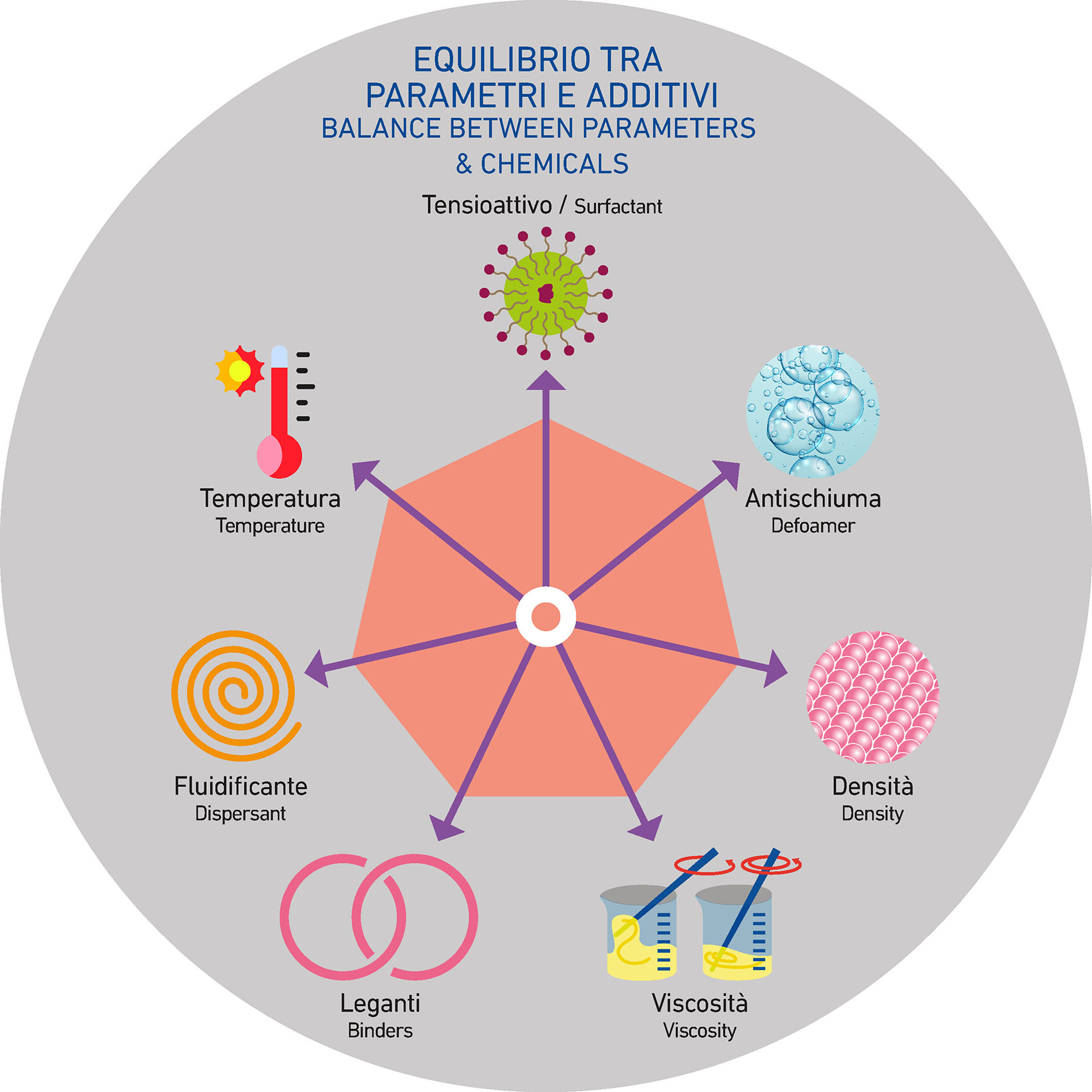
In the case of cratering phenomena, as we said, the most important parameters are:
- Density
- Viscosity
- Temperature
- Surfactants
- Defoamers
- Dispersants
- Binders

