#34 Sodium silicate & deflocculation of ceramic mixtures
- Sodium silicate: identikit
- From a chemical point of view
- The deflocculation:
- Cation exchange
- Complexation
1. Sodium silicate: identikit
Sodium silicate is an inorganic compound that at room temperature is at solid state. It is a chemical compound with many different uses: as insecticide and fungicide, as fireproof substance in construction field, as dyer for cotton in textile industry or in the field of art where is used to protect wall paintings.
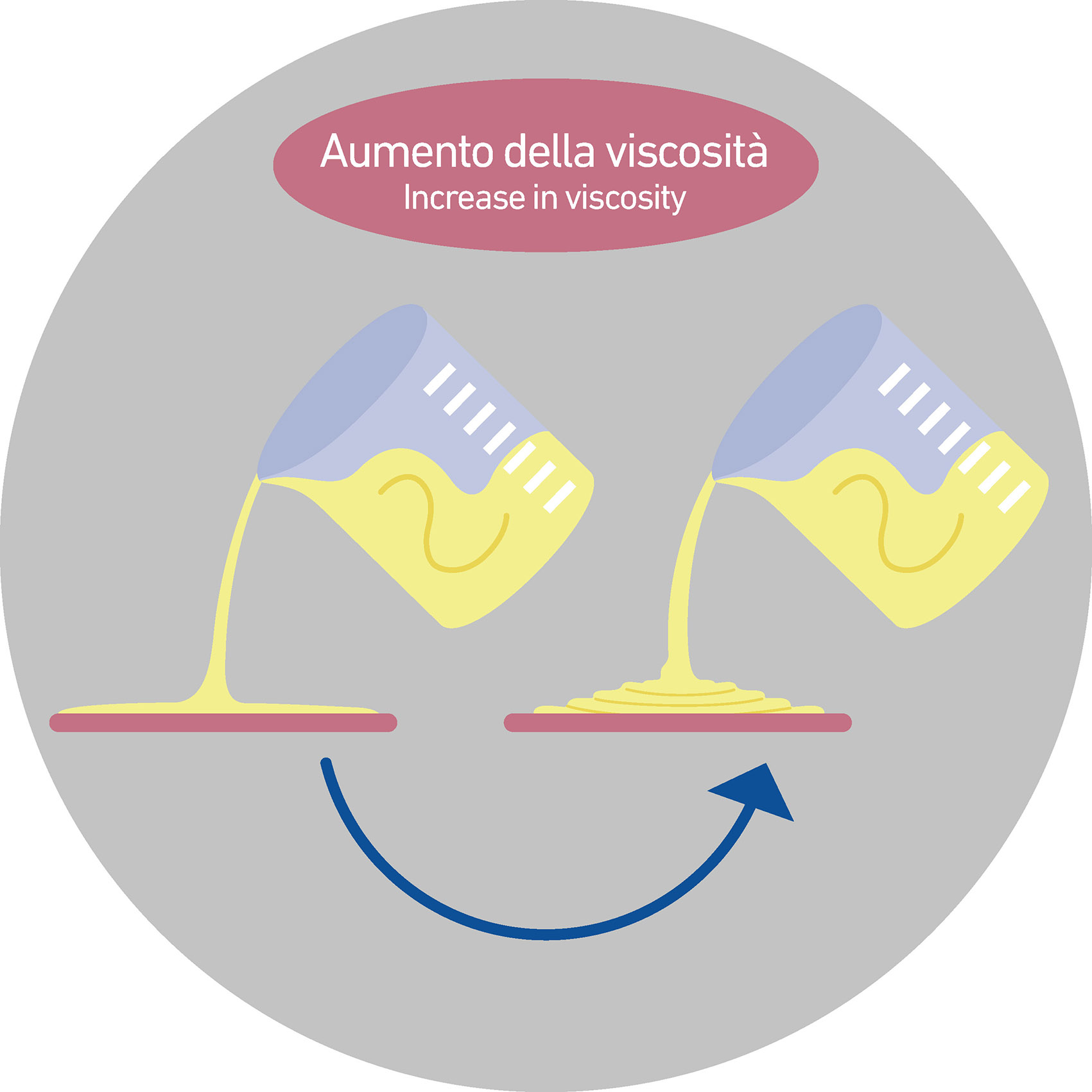
In ceramics, sodium silicate is very effective within ceramic mixtures, Among the several effects it develops, it plays an important role in terms of fluidization.
As dispersant for ceramic mixture (chemicals able to decrease viscosity values of a fluid), it allows you to reduce the amounts of organic compounds within the formula that, due their decomposition, are the basis of those degassing phenomena that may arise during the firing stage.
These phenomena, in fact, if not correctly managed, can lead to porosities within the tile, therefore producing serious technical and aesthetic problems.

The reduction of the organic matter promoted by the presence of sodium silicate, acts also in regards to the reduction of those degassing phenomena that may lead to the formation of the so-called black core. That is a localized area marked by a brown/black (or even white) color, clearly visible in the thickness of the ceramic tile that it is usually sign of an improper oxidation and that it can be considered as an important aesthetic defect.

It may seem obvious to underline it but sodium silicate, since it is siliceous based, is marked by the ability to facilitate the formation of glass, thus triggering that process of vitrification and sintering that is the basis of the formation of porcelain stoneware.
SINTERING PROCESS
Process of compacting and forming a solid mass of material by heat or pressure without melting it to the point of liquefaction. It is used to produce material with specific features that would not be effective if created with a different system. It is a densification process that at the same time provides a significant decrease of the porosity.
2. From a chemical point of view
Sodium silicate (also called water glass) is a polymerized substance in aqueous suspension. It is a polymeric chain of silicate in anionic form, where the cation is sodium.
POLYMER
A polymer is a macro molecule with a high molecular weight. It consists on a long chain that can be provided with many branches. The structure is formed by several basic units: these are single molecules, monomers, that can be described as pearls of a pearl necklace. They may be aggregated in groups of two, three, four…up to hundreds (high polymers). Every monomer is linked to the others by a chemical bonding: the polymer can be polymerised using solvents able to break the chains.
ION
Atom or molecule electrically charged. An atom – or a molecule or even a group of bound atoms bound – that gives up or gains one or more electrons, it turns into ion. The ions can have both a positive or negative charge: the first one is called CATIONS, the second ANIONS.
3. The deflocculation
Sodium silicate, thanks to its features, is able to cover the main mechanisms of fluidization process.
CATION EXCHANGE
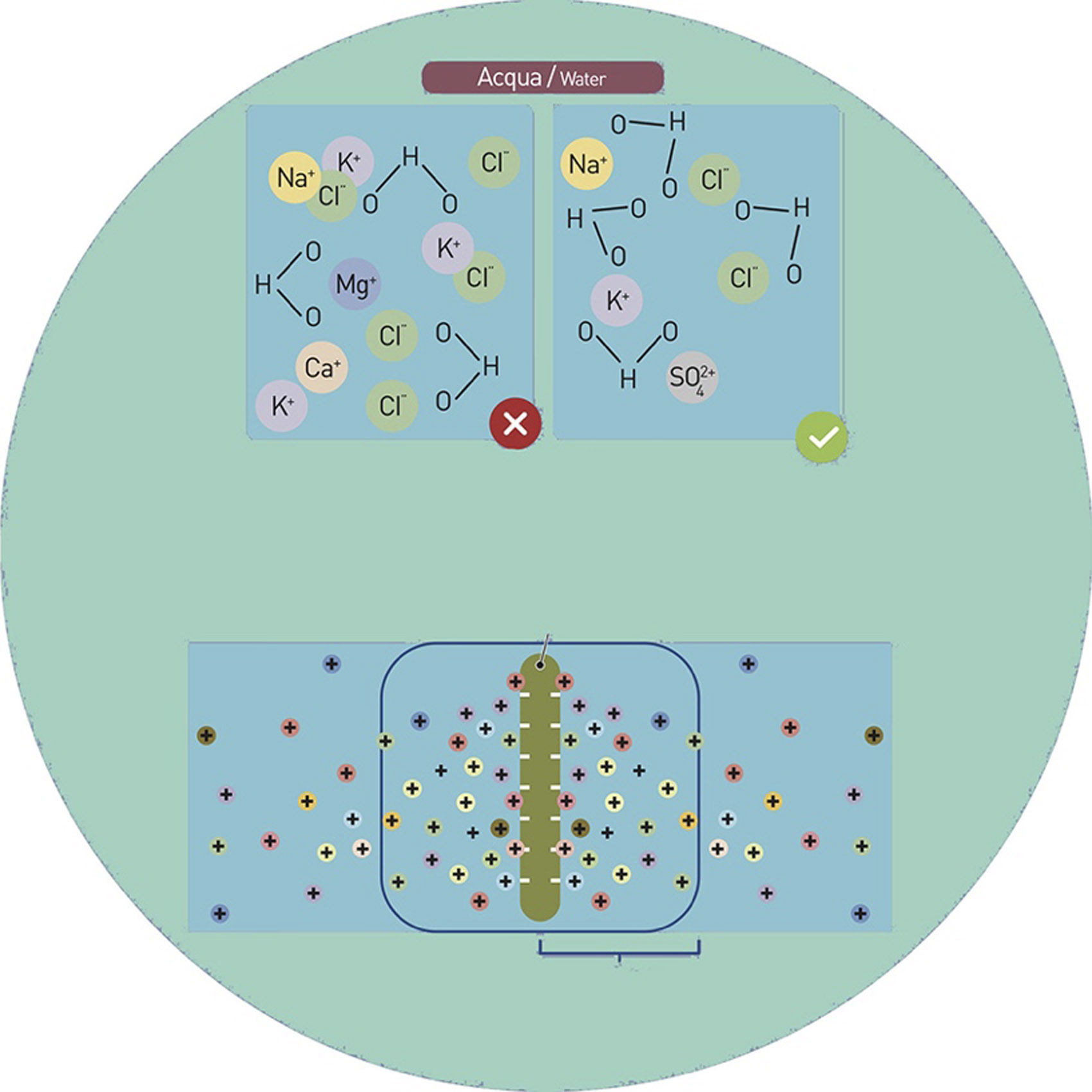
Sodium, as a monovalent cation, produces cation exchange with the bivalent and trivalent cations that are within the solution. This exchange is a phenomenon that leads to a reduction of the agglomeration phenomena that are the basis of flocculation and that, thanks to the opposite process, reduces the viscosity values of the system.
The use of sodium (monovalent cations) allows the replacement of the multivalent positive charges present on the clays with weaker charges. This specific type of cation exchange reduces the positive charge on the clay micelles without neutralizing the negative charges. This results in a reduction in agglomeration and in a decrease in the viscosity of the system.
COMPLEXATION
Again, sodium silicate has a strategic role.
Complexing agents consist of particular chemical molecules (sodium salts) equipped with functional groups containing atoms that provide the system with a very negative electronic charge.
When added in the ceramic mixture, the complexing agents release sodium (monovalent cation) preferentially attracting multivalent cations. The result of this electrostatic process facilitates cation exchange with a consequent increase in the distance between the particles and a decrease in viscosity, containing if not to eliminating the flocculation phenomenon.
IS SODIUM SILICATE ENOUGH?
Although the action of sodium silicate is relevant, it is not completely effective in avoiding flocculation phenomena. Its presence must go hand in hand with the use of synthetic polymers able to enhance its performance. In this regard, it is important to study, develop and use dispersants able to meet all the needs of each production line.

