#16 Black core: origins & solutions
- Definition & introduction
- The origins of the problem
- Interventions & resolutive actions :
- Production line set-up
- Chemical actions
1. Definition and introduction
The black core is a localized area marked by a brown/black (or even white) color, clearly visible in the thickness of the ceramic tile.
Its presence is usually sign of an improper oxidation but it has not to be considered as a defect that compromises the product functionality (unless the black core comes with a swelling).
Note
Oxidation is a reaction by which an element or compound loses electrons, giving them to another substance. More generally, it’s a combination reaction with oxygen.

According to that definition, it is important to underline that swelling phenomena that develop in the thickness of the tile are now increasingly unusual. In fact, the inorganic raw materials currently used in ceramic production are marked – to a large extent – by a low content of organic (that is the main responsible for the problem).
In other words, the content of organic carbonaceous products is normally very little.

However, the increasingly higher densities of ceramic mixtures – that positively affect the industrial productivity – can be a problem for the proper expulsion (that is evaporation) of the organic materials during the pre-firing phase. The organic material, since it remains within the ceramic body, is the most important pre-condition of the critical issue.

The black core is only an aesthetic defect: it has been proven, in fact, that in many cases its presence may even increase the mechanical resistance of the ceramic material.
Even if black core does not affect the technical features of porcelain stoneware, it must be carefully managed because of the increasingly different intended use of the ceramics.
Since tiles (especially big slabs) are now used not only for coverings (wall and floor) but also for kitchen countertops, tables, and more generally for furniture, the section/thickness of the material can be visible and therefore it must not be marked by any aesthetic defects.

2. The origins of the problem
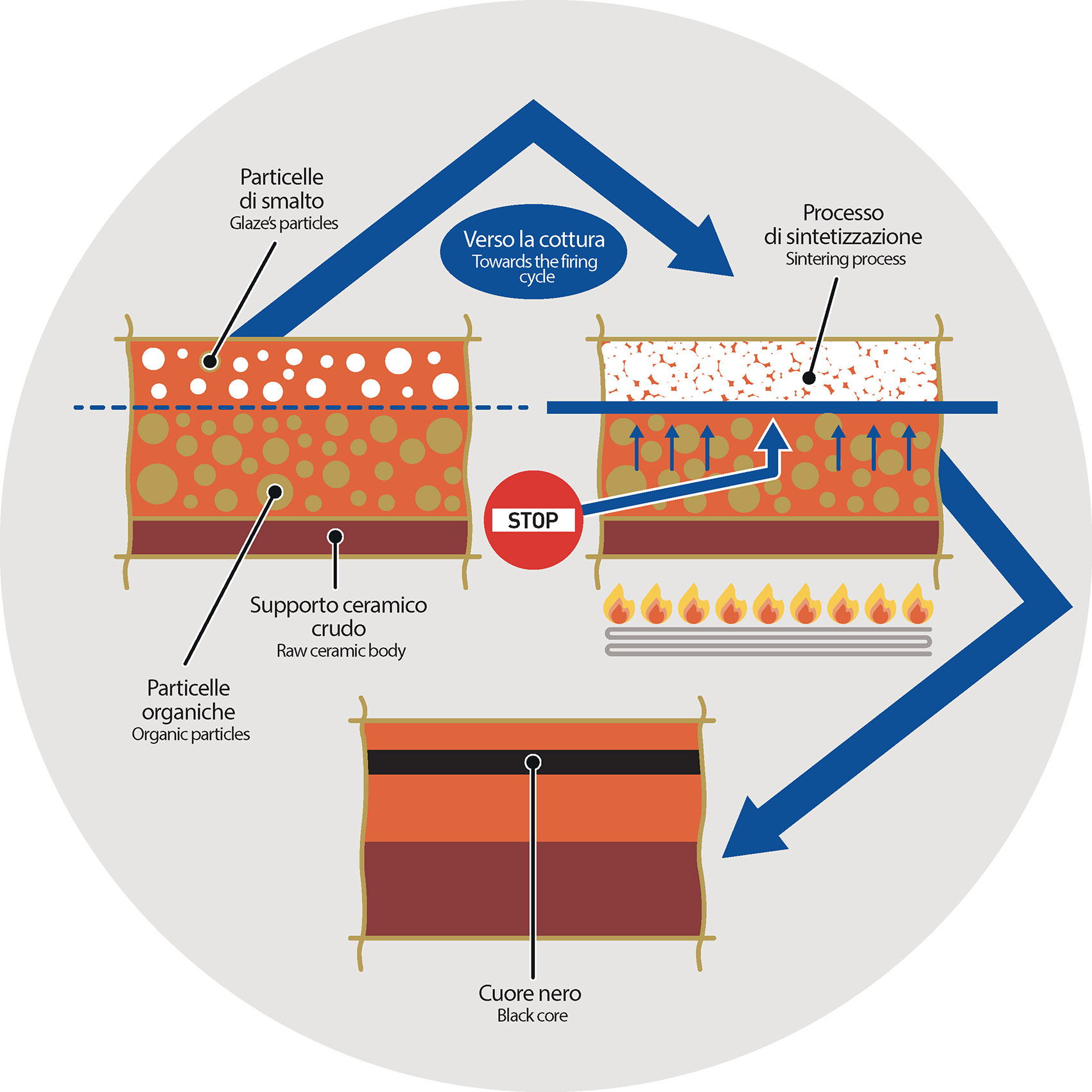
They are usually due to some components (both organic and inorganic) that before the sintering process do not find in the central area of the tile the proper chemical-physical conditions to uniformly react such as in the other parts of the ceramic body.
This different kind of reaction can keep (hold) the organic materials inside the tile cause organic substances to be stuck inside the tile even after the sintering process.
Sintering process: process of compacting and forming a solid mass of material by heat or pressure without melting it to the point of liquefaction. The lack of expulsion of the organic materials is mainly due to the very short firing times.
3. Interventions and resolutive actions
a) PRODUCTION LINE SET-UP
Even before using chemicals, it is possible to act in advance by refining and adjusting the set-up of the production line. Following here the most representative.
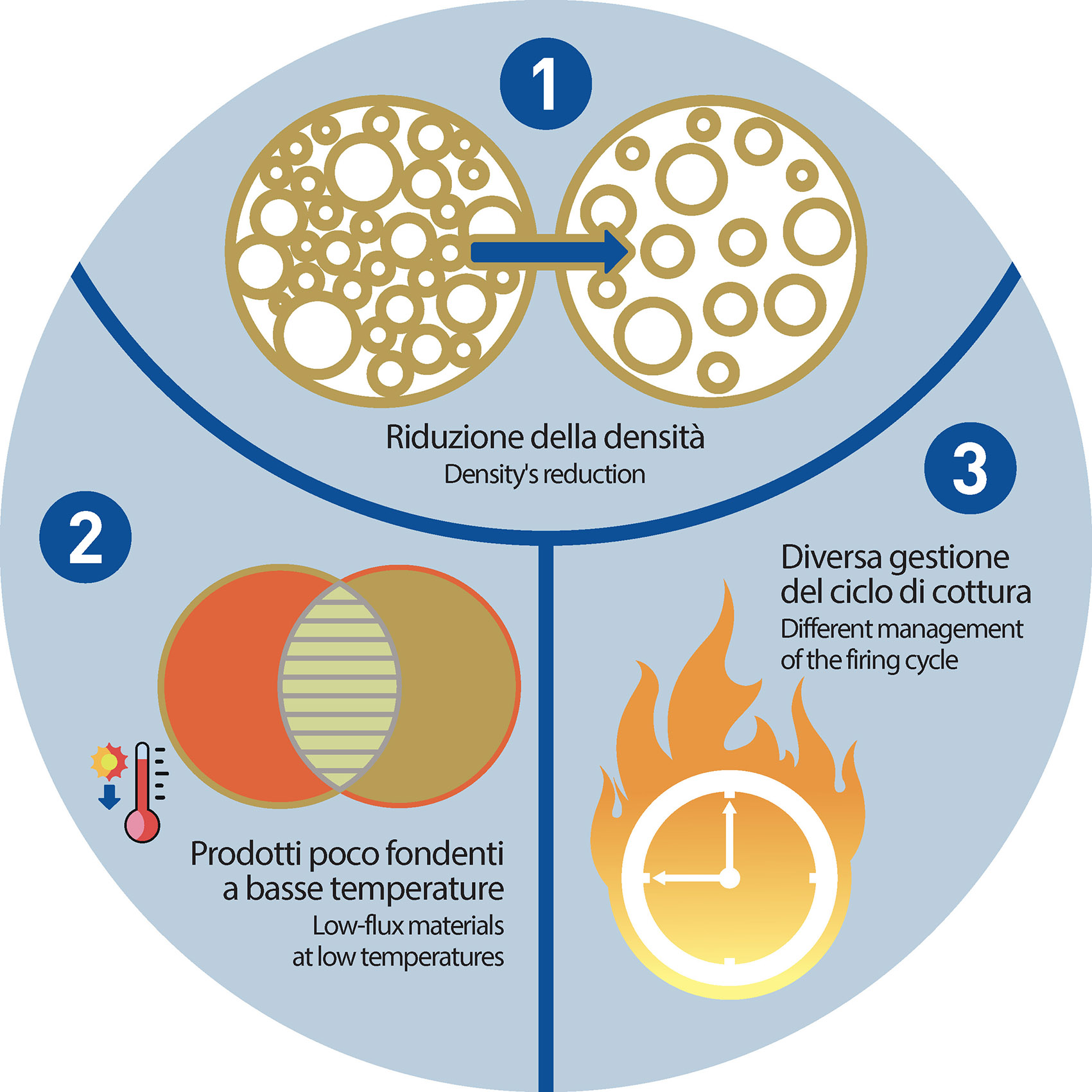
- To slightly decreasethe density values of raw tiles. There are many options to it, such as – for example – a reduction of the pressure on the tile during the forming phase
- To avoid along the glazing line the use of products marked by a too low melting point, so as they do not merge before the organic components had the time to properly leave by evaporation the ceramic body (i.e. before the sintering process)
- To differently manage the firing cycle (increasing, for example, the times)
b) CHEMICAL ACTIONS
From the chemical point of view, it is possible to use specific chemicals – that can be added both at the grinding stage and inside the barbottina – that bring into the ceramic body molecules able to provide oxygen to the organic substances and so to promote a better degassing process during the pre-firing phase. Oxygen promotes the formation of organic molecules that, at pre-firing temperatures, leave the tile in gas form in proper times, reducing or even avoiding the problem.
More precisely, the oxygen provided by this category of chemicals is able to turn some of the organic molecules of the ceramic mixture into more elementary forms, until the formation – at best – of carbon monoxide or carbon dioxide, gas capable of evaporating from the tile before sintering.
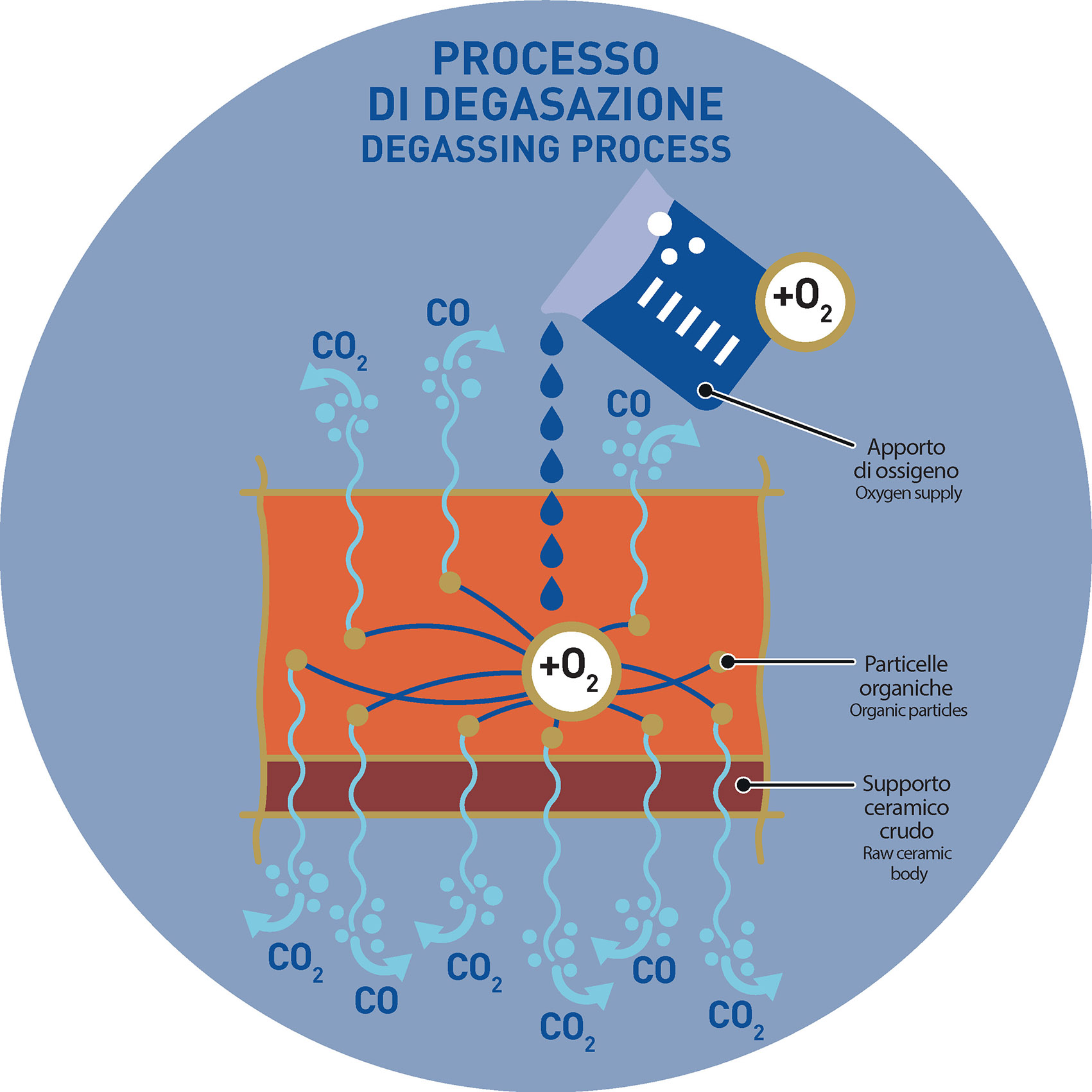
The black core, especially in the most colored mixtures, may sometimes come from pigments that are marked by an oxidative state that is different from what they should have to properly develop the required color. In oxygen deficiency (kiln reducing atmosphere) the pigment can move from an oxidized form to a more reduced form of oxidation: a form in which the pigment could change its color.
The OXIDATIVE STATE of an atom defines the numbers of electrons in the atom’s outermost electronic band. In other words, the kiln’s oxidizing or reducing atmosphere during the firing phase is essential to ensure that the colored ceramic mixture develop, after firing the surface color required by the project (maximum color rendering). Under improper conditions, in fact, the pigment may unevenly change the color of the tile’s surface.
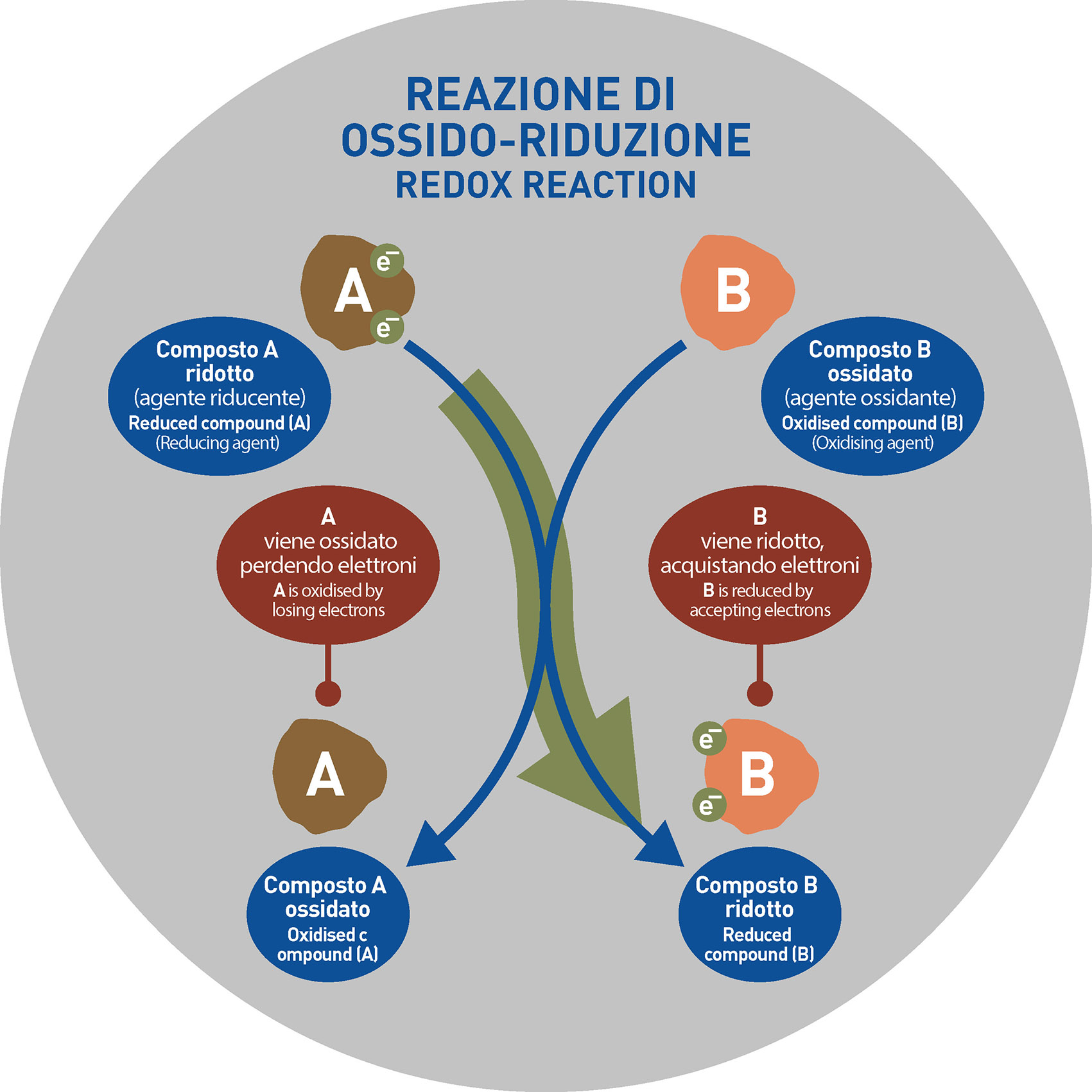
It is clear that the deeper parts of the tile are the less involved in the oxide-reduction process because they are not close to the surface and less in contact with the atmosphere within the kiln: the oxidizing atmosphere of the kiln does not reach the deepest parts of the tile.
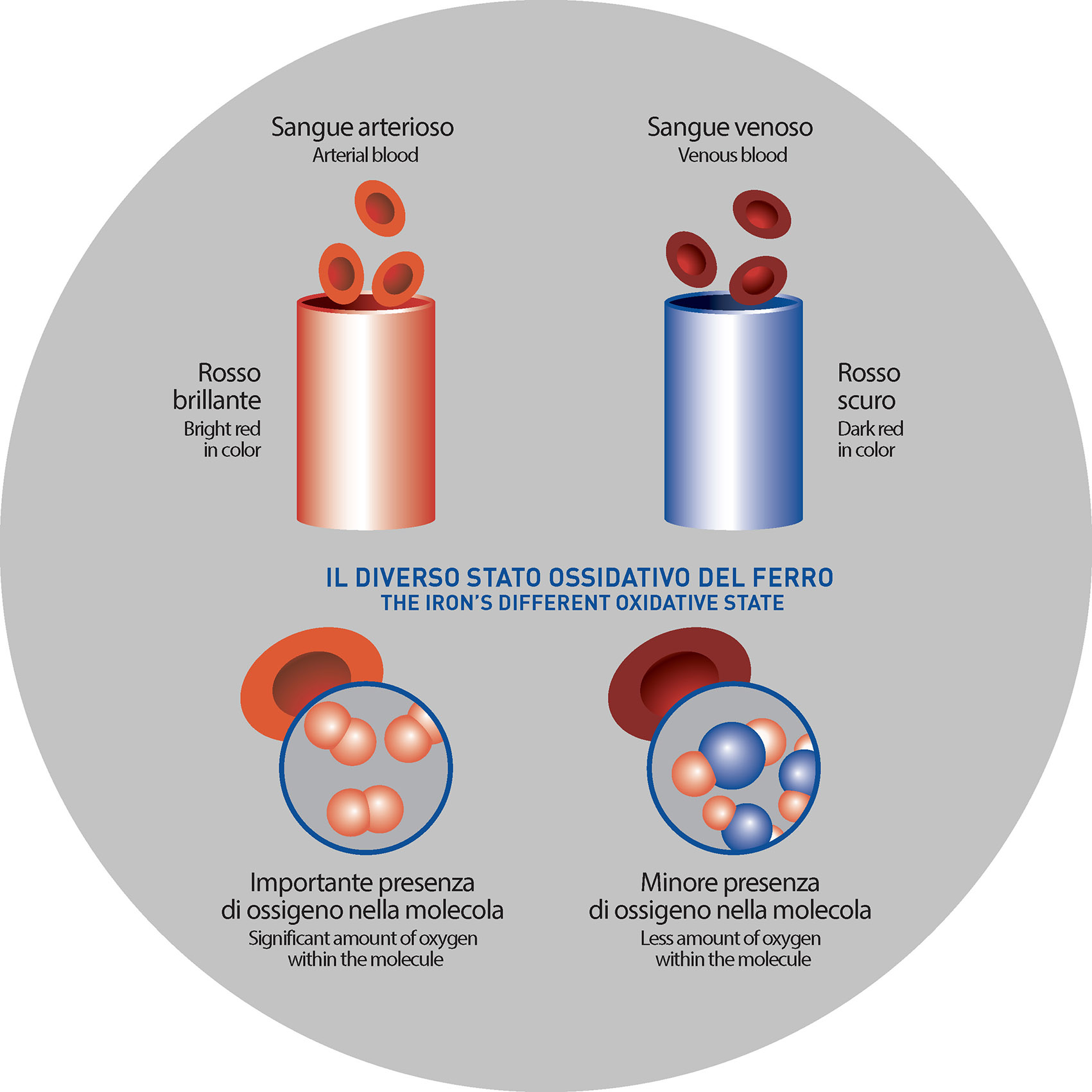
A CLEAR EXAMPLE: THE BLOOD
Even the blood, according to the level of oxidation (or oxidative state) of the iron, can significantly change its color:
- Arterial blood is rich in oxygen and so the iron is marked by an oxidative state that makes the blood bright red
- Venous blood, poor in oxygen and characterized by a different oxidative state of the iron, is dark red and dull
Going back to ceramics, in all those cases where the black core is due to a different state of oxidation of the pigment, it is possible to use appropriate chemicals able to act and change the oxidation state of the pigment by standardizing its tone to the other parts of the mass. In other words, it is possible to act by developing real REDOX reactions.