#13 Superficial halos formation during the drying process
- Residual humidity & clays’ performance
- Improper drying process
- The source of the problem
- Problem-solving actions:
- The dryer
- Dispersants for ceramic mixture
- Process waters
- The ceramic mixture formulation
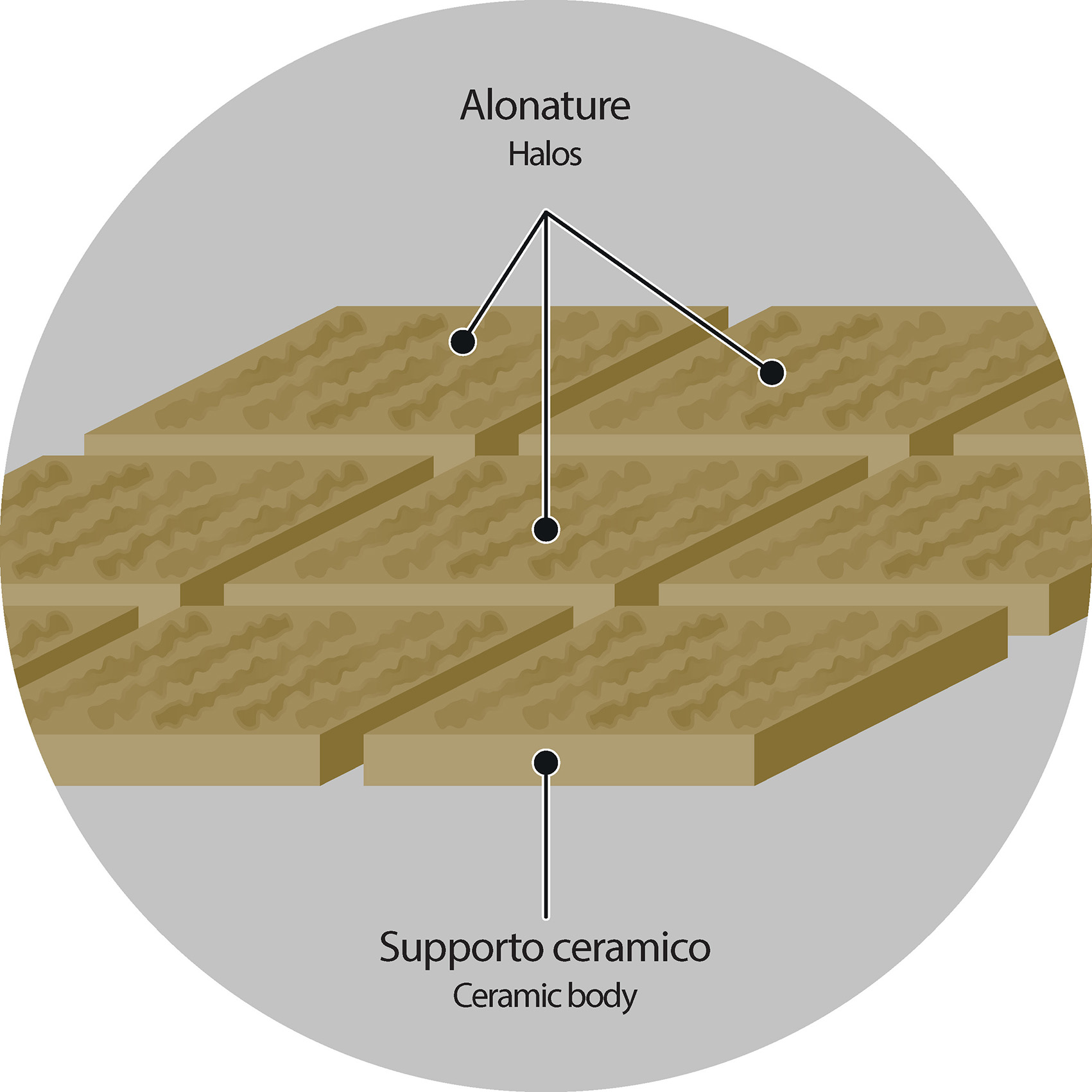
1. Residual humidity and clay’s performance
As we know, after the atomization process and during the forming stage, the ceramic mixture undergoes to very high pressures that can be more than 400 Kg/cm2 and that are required to correctly shape the ceramic body.
The raw tile, even after pressing, contains a water amount (that is residual humidity) that is about 5/7%.
Its presence is fundamental to provide the clays with good plasticity level that ensure the proper development of the process under the presses. However, if the presence of residual humidity allows on the one hand to give shape to the ceramic support, on the other hand it negatively affects the raw tile’s mechanical features that are necessary for subsequent applications.
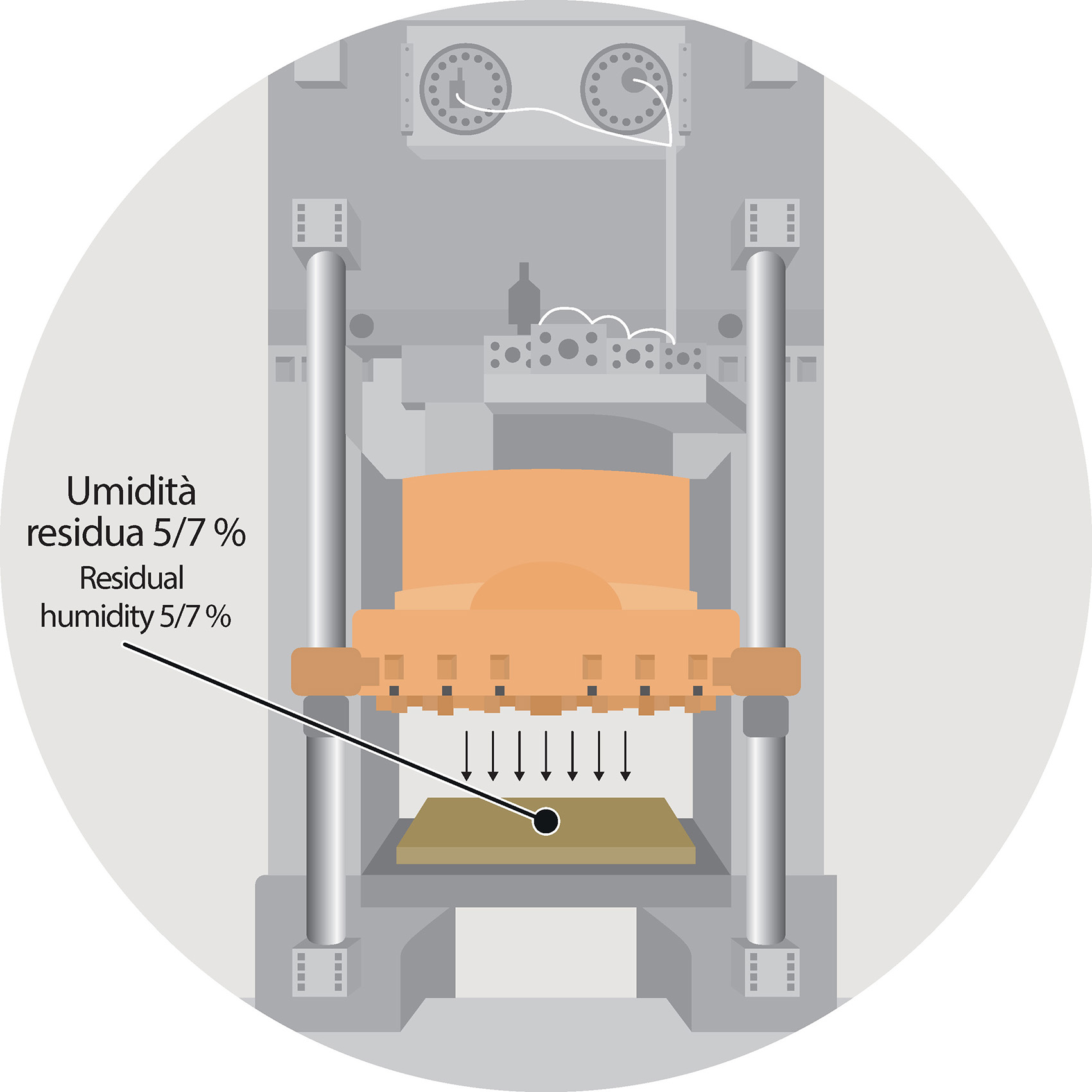
To increase the mechanical resistance of the raw tile it is therefore necessary the elimination of the residual humidity from the system by means of an appropriate drying cycle, developing a gradual expulsion of the water and so preventing the stresses that can give rise to defects. Even at this stage of the process.
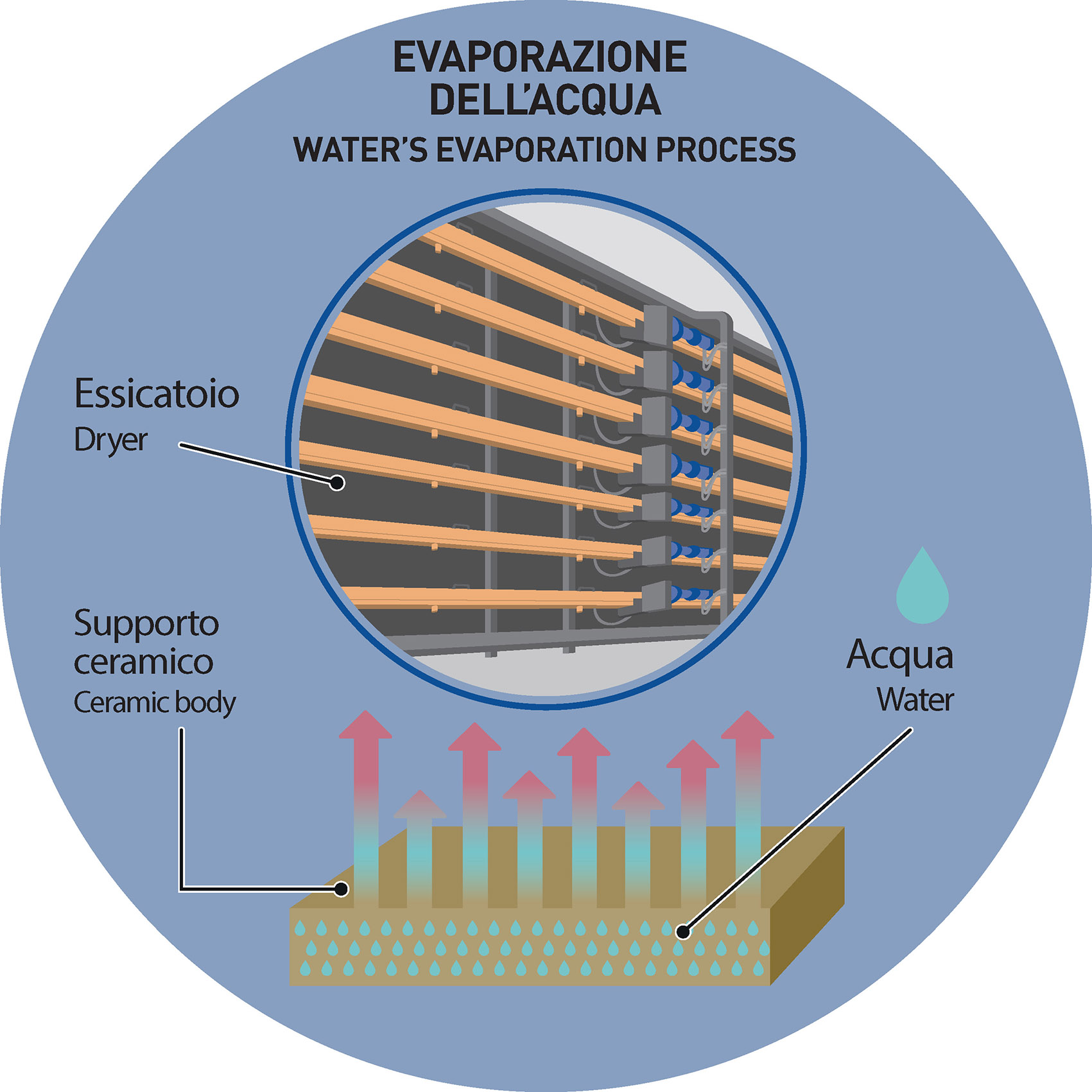
More specifically: the water molecules, thanks to the drying process, must progressively migrate from the center of the ceramic body towards the outer layers and then evaporate, allowing the system to bind and minimize the consequences of possible stresses.
2. Improper drying process
During the drying process there are no problems due to the tile’s shrinkage when:
- The atomized clay powder complies with the formulation
- The atomized clay powder contains the proper amount of residual humidity
- The pressing action is uniform and without anomalies
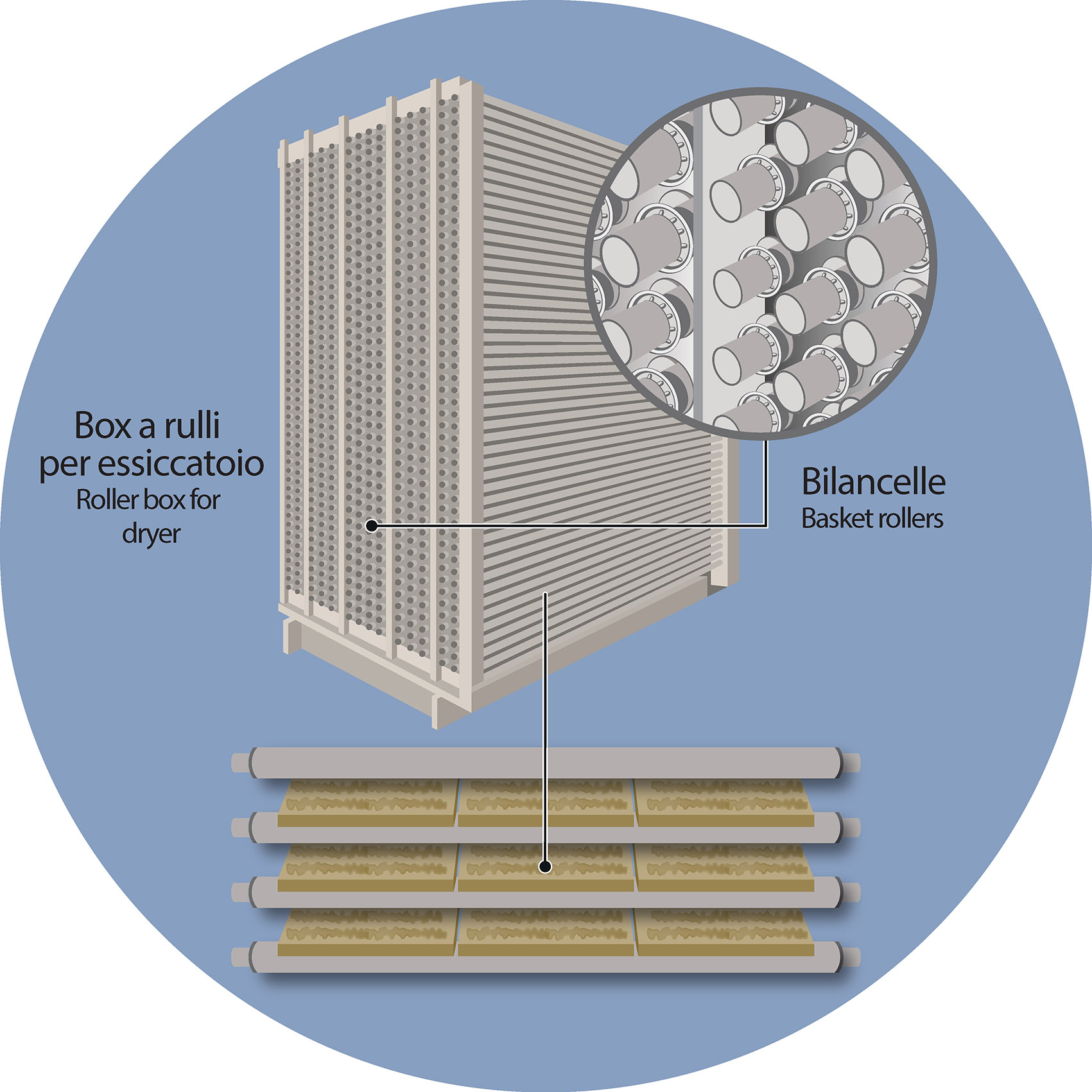
The drying process is held by means of dryers in which the raw tiles are placed on metal supports (roller stands) for a preset time, until the presence of residual humidity is reduced to values close to 0%.
The progressive heating to which the ceramic body is subjected inside the dryers produces the aforementioned migration of water molecules that therefore leave the ceramic body by evaporating.
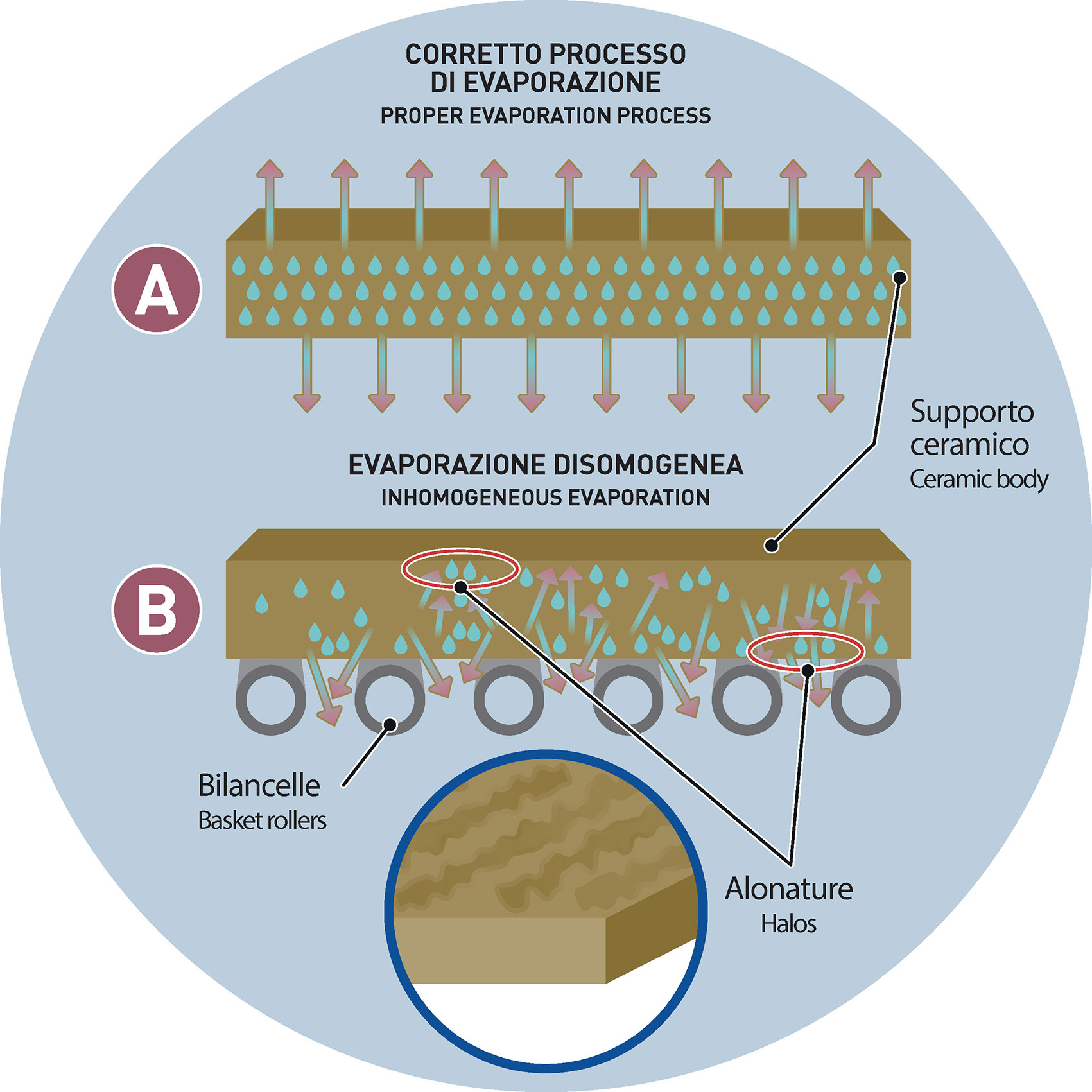
This process, however, may in some cases not be completely homogeneous over the entire surface of the tile. More precisely: the parts of the ceramic body that rest on the basket rollers may be marked by different evaporation times (and modes) compared to those areas of the tile that are not in contact with the metal parts of the dryer.
The different evaporation mode that takes place in both the lower and upper parts of the tile, in some rare cases produces longitudinal bands/stripes that may remain visible even after the drying cycle.
Basically, these are brilliant halos that, despite the subsequent applications of inks and glazes, may persist and be visible even after firing.
The problem specially arises in case of technical ceramics production or in case of non-massive applications along the glazing line.
3. The source of the problem
What is the reason of this uneven degrees of gloss (or brilliance)?
The reason must be found in the presence of some particular ANIONS and CATIONS (particular chemical species formed by one or more atoms) that freely move within the mixture and that during the drying phase are carried by water’s migration process.
Since they do not evaporate, these kinds of anions / cations tend to concentrate unevenly on the surface, in some areas rather than others: the areas with a higher concentration show a different degree of gloss.
In general, all anions and cations that do not form insoluble salts within the system (or that have low mobility due to their conformation) do not chemically bind to the mixture and tend to move together with the water towards the surface.
Who promotes the presence of this type of anions/cations?
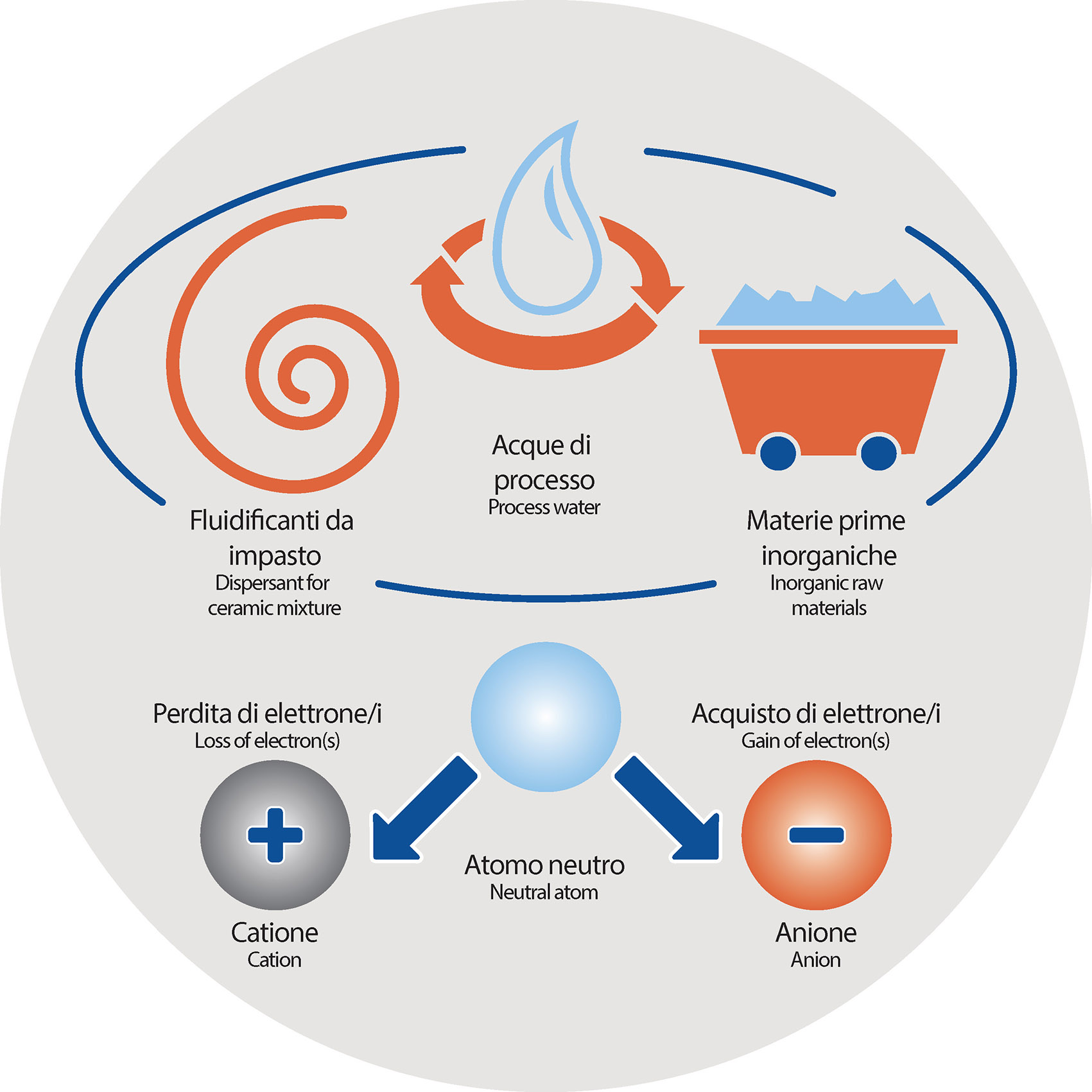
Without oversimplify, among the various sources of the problem three are the most relevant:
- Dispersant for ceramic mixture (sodium silicate-based)
- Process waters
- Ceramic mixture’s inorganic raw materials
The type of clays and their different plasticity can positively or negatively affect the result, according to their ability to retain or regulate the water migration (and therefore the movement of ions/cations) to the surface.
4. Problem-solving actions
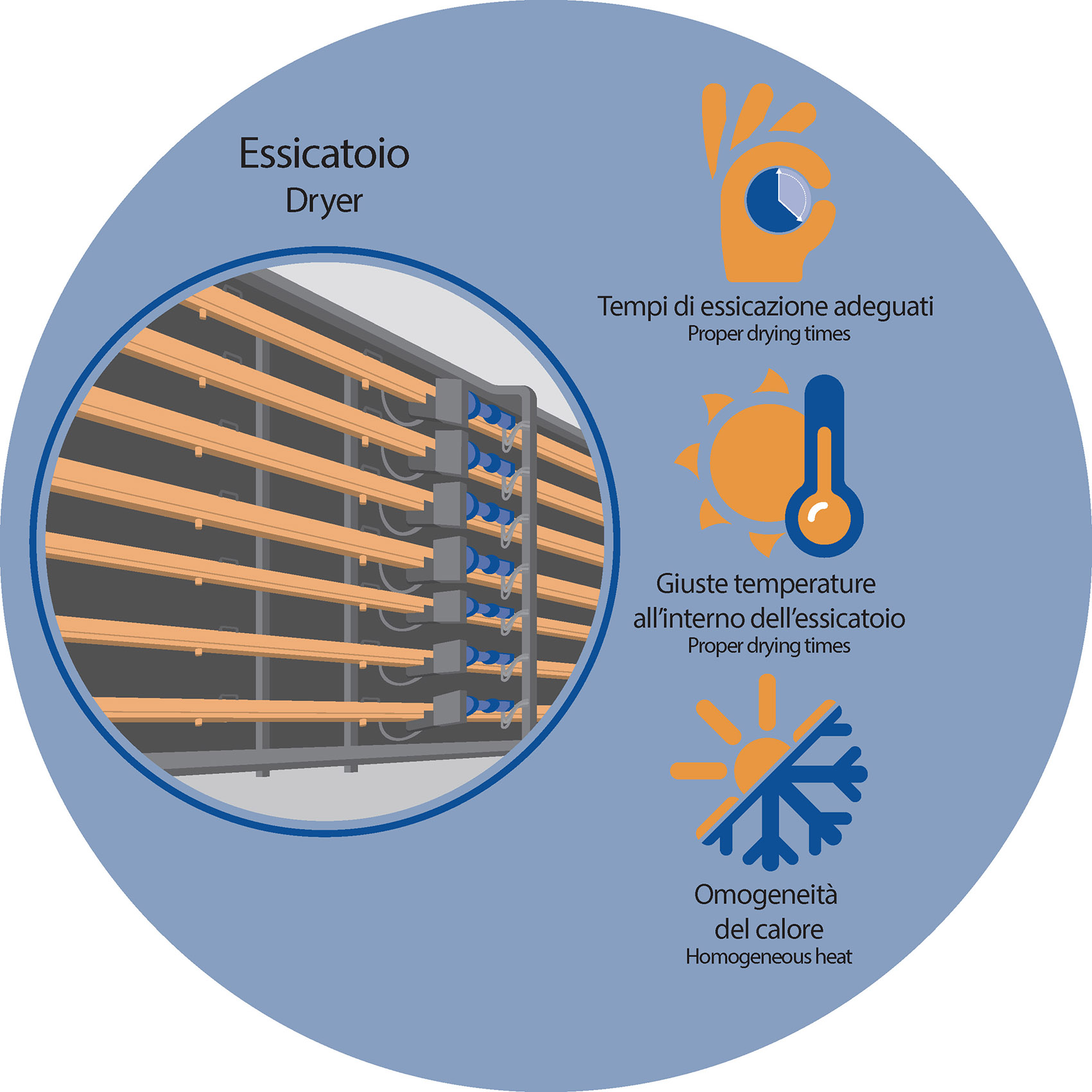
A. THE DRYER
To prevent the problem, it is firstly important to be sure about the proper developing of the drying cycle in terms of times, temperatures, and homogeneity of the heat inside the dryer. The good set-up of the plant is, in this regard, a pre-condition to prevent the problem.
B. DISPERSANTS FOR CERAMIC MIXTURE, SODIUM SILICATE AND WATER SOLUBILITY: A STRATEGIC CHOICE
If the issue persists even after the dryer’s perfect set-up, a chemical intervention is required to eliminate the problem at its roots.
From this point of view, it is necessary, for example, to check the dispersants involved in the grinding phase: most of these agents, in fact, are based on sodium silicate, a chemical compound completely soluble in water that in several cases may be critical.
The choice of thendispersant is therefore crucial: selecting a product marked by a low solubility means that the active ingredients it contains (i.e. ions and cations) – in the presence of a residual humidity content of 5/7% – are only partially transported by water during its migration and evaporation process.
NOTE
It is possible to formulate dispersants using special polymers or molecules capable of limiting the migration of ions to the surface.
C. PROCESS WATERS
It is also important to pay attention to the chemical composition of process waters. After a careful study of their characteristics, it is necessary to check possible excessive amount of those particular anions and cations that can be responsible for the halos.

For these reasons, it is therefore advisable to limit the presence of anions and cations of inorganic origin that tend to create soluble salts within the process waters.
Although the electrical conductivity of water is not a parameter able to precisely identify and define the type of ions within the water, the check of its values can give indications about the possible massive presence of ions.
If so, it is usually possible to take action on the process water, by adding mains or ground water to decrease the activity of the ions.
D. THE CERAMIC MIXTURE FORMULATION
Although this happens rarely, acting on the formulation of the mixture could finally be a useful way to contain the problem.
For example, it could be possible to replace, introduce or increase some kind of clays capable of retaining more water or at least of slow down and standardize the ions and cations’ migration.
For example, more plastic clays (such as bentonites) absorb and retain much more water and tend to release it more slowly, promoting a regulated migration of water and ions.

