#49 Suspending agents: nature, functions & chemical-physical actions
- Introduction: identikit
- Suspending actions: points of intervention:
- a. Viscosity & rheological behaviour
- b. Electrostatic repulsion
- c. Concentration: liquid-to-solid ratio
- Suspending action and type of suspensions:
- a. Barbottine
- b. Glaze suspensions
- c. Grit suspensions
1. Introduction: identikit
Suspending agents fall under a broader category of additives whose primary function is to enhance the internal stability of ceramic suspensions—whether slips, engobes, glazes, or grits—by preventing sedimentation phenomena.
Available in both liquid and solid form, these products work within the suspension to maintain what could be described as a proper internal balance.
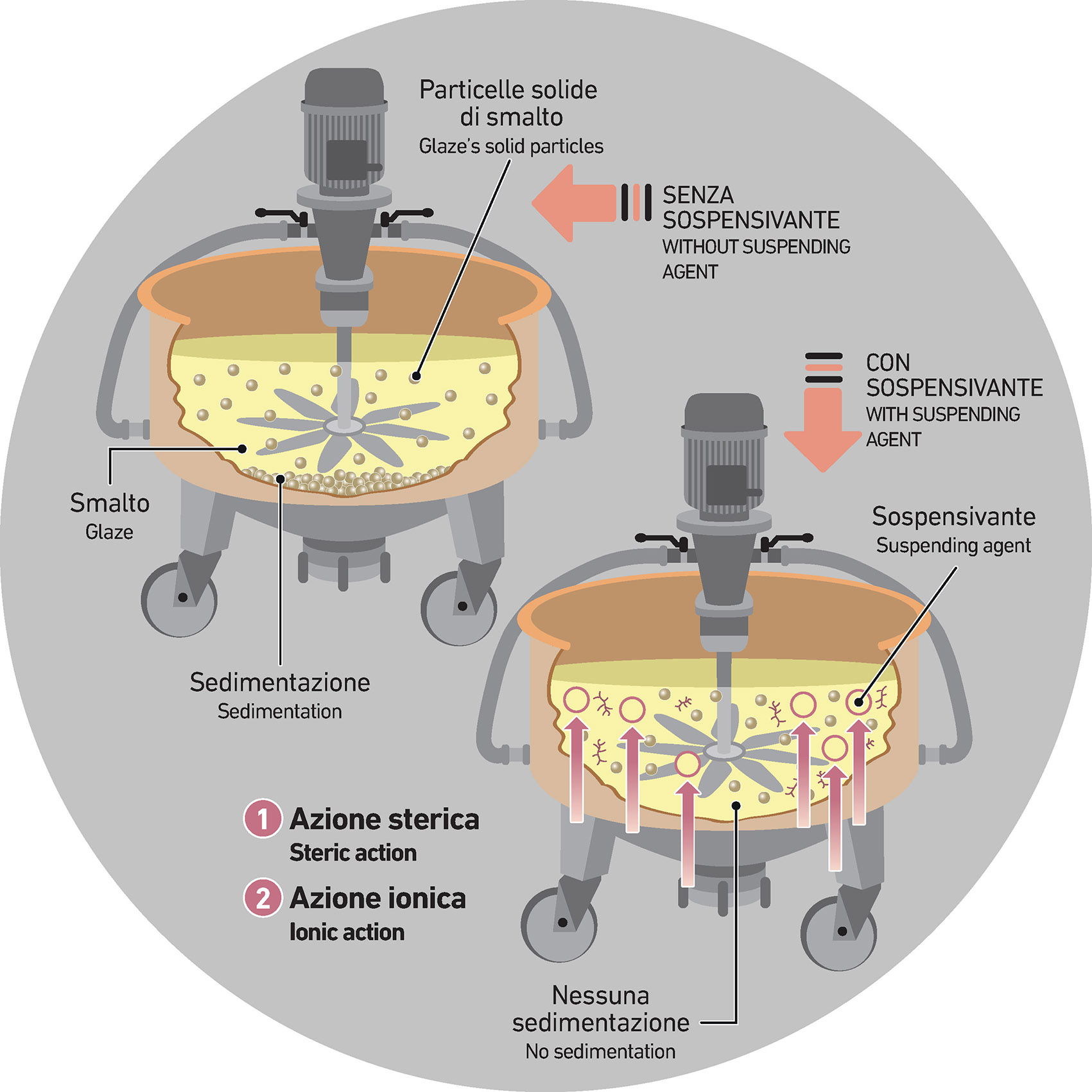
Suspending agents represent a broad and varied family of additives whose role is to help keep ceramic suspensions—be they slips, engobes, glazes, or grits—stable and homogeneous over time.
While their underlying mechanisms may differ, involving a range of chemical and physical principles, their common goal is to prevent solid particles from settling and disrupting the balance of the mixture.
These products come in both liquid and solid forms and, rather than belonging to a single chemical class, they span across multiple additive types. In many cases, substances designed for entirely different primary functions can also provide a stabilizing effect on the suspension, contributing to its overall performance.
The choice of suspending agent, however, is never one-size-fits-all. Each ceramic suspension has its own unique profile—solid content, particle size, viscosity, and more—and these differences must be carefully considered when formulating the right additive package. On top of that, the specific production technology being used adds another layer of complexity, influencing how the suspension behaves and which additives are most effective.
In short, the list of functions these additives can perform is long and often application-dependent. It’s difficult to summarize them neatly, but some key parameters can help guide selection and formulation. Ultimately, the biggest challenge remains gravity: the natural force pulling solid particles to the bottom of the suspension, which—if left unchecked—can make the mixture unworkable and compromise the quality of the final product.
2. Suspending actions: points of intervention
There are several ways to achieve effective particle suspension, but in general, we can identify three main approaches that help keep solids evenly dispersed within a ceramic system.
A) VISCOSITY & RHEOLOGICAL BEHAVIOUR
One of the most common methods involves the use of specific types of polymer-based materials.
These additives influence both the viscosity and the rheological (flow) properties of the suspension, helping to prevent particles from settling too quickly.
What happens at the microscopic level?
That depends largely on the specific additive being used.
There’s a wide range of products on the market generally referred to as rheology modifiers. These can be natural or synthetic, but they often share some common traits: complex, often branched or 3D molecular structures and highly polar functional groups.
When mixed into water, some of these polymers swell and uncoil, forming a loose, three-dimensional net inside the fluid. Thanks to their polar groups, the polymer chains interact with each other, creating a microstructure that slows down the movement of solid particles.
This structure, however, isn’t permanent. It breaks down easily when the suspension is stirred or put into motion, and reforms once the system is at rest. This reversible behavior is known as thixotropy in rheology.
In simple terms, when the suspension is still, the presence of the polymer increases its viscosity. This thicker fluid makes it harder for particles to move, especially downward. As a result, sedimentation slows down significantly—even though gravity, of course, can’t be stopped entirely.
There are also other types of polymers that raise viscosity without forming any kind of internal structure. These are typically Newtonian fluids with very high inherent viscosity. When added to a suspension, they increase its thickness across the board.
As mentioned, the higher the viscosity, the greater the resistance to movement—not just for the fluid itself, but also for the solid particles within it. The more viscous the medium, the harder it is for the particles to sink, which helps the suspension remain stable for longer.
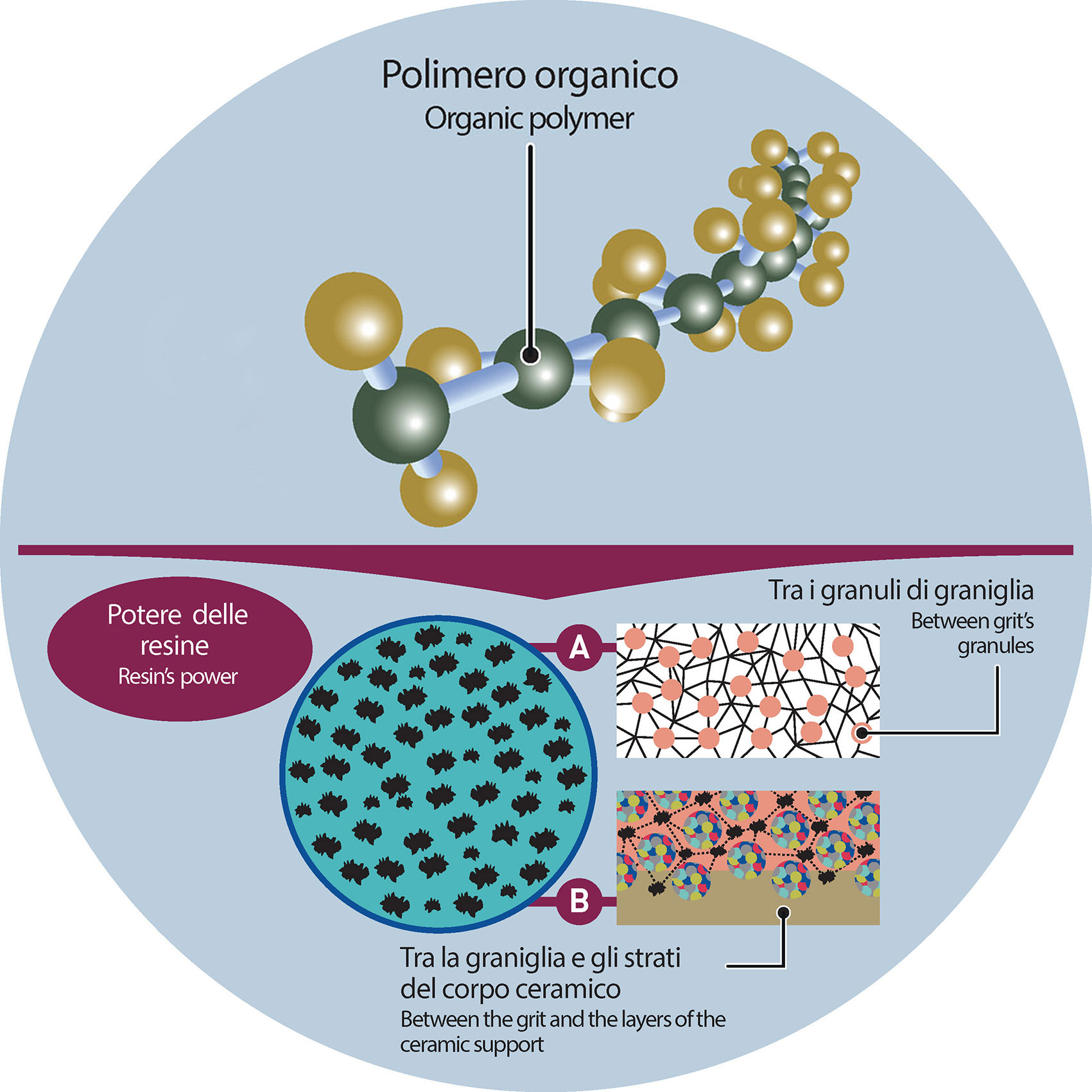
FOCUS: POLYMERS
A polymer is a macromolecule—that is, a very large molecule made up of many smaller repeating units called monomers. These monomers are linked together by covalent bonds, forming long chains or even complex three-dimensional net. What makes polymers unique is precisely this chain-like structure, which gives them physical and chemical properties that are often very different from the smaller molecules they originate from.
By altering the structure of the chain, the type of monomers used, or the conditions under which the polymer is formed, it’s possible to fine-tune the final material’s behaviour. Some of the key factors that influence how a polymer performs include:
- Molecular weight, which affects viscosity, mechanical strength, and melting point.
- Degree of polymerization, or the number of monomer units in a single chain—this determines the overall chain length.
- Branching, meaning the presence of side chains, which impacts both the mechanical and thermal properties of the material.
While the word polymer might bring to mind common materials like polyethylene used in packaging, or rubber in tires, there is a whole category of water-soluble polymers that are of great technological interest. These are used in a wide range of applications: water treatment, biomedical formulations, cosmetics, detergents, and—more recently and increasingly—in ceramic manufacturing.
B) ELECTROSTATIC REPULSION
One effective way to stabilize a ceramic suspension is by using additives that influence the electrostatic repulsion between particles. These additives—often electrolytes—work by modifying the electrical double layer that surrounds each particle. By increasing the concentration of ions in the system, they can fine-tune the interactions between particles and help maintain a stable suspension under specific conditions.
The electrical double layer is a microscopic structure that forms at the interface between a solid surface and the surrounding fluid. It consists of two zones: a tightly bound inner layer where ions are adsorbed directly onto the particle surface, and a looser outer layer made up of free ions distributed in the surrounding liquid, influenced by both electrical and thermal forces.
What keeps the particles apart—and therefore the suspension stable—is the electrostatic repulsion generated by this double layer. The thickness and density of the double layer determine how strongly particles repel each other. A well-developed double layer helps prevent particles from coming too close, thus reducing the risk of clumping or settling. However, the presence of multivalent ions—like calcium or magnesium—can compress the double layer, making it thinner. When this happens, the repulsive force between particles drops, making them more likely to aggregate. Once particles start forming clusters, the suspension can thicken or even become unstable, compromising its processability.
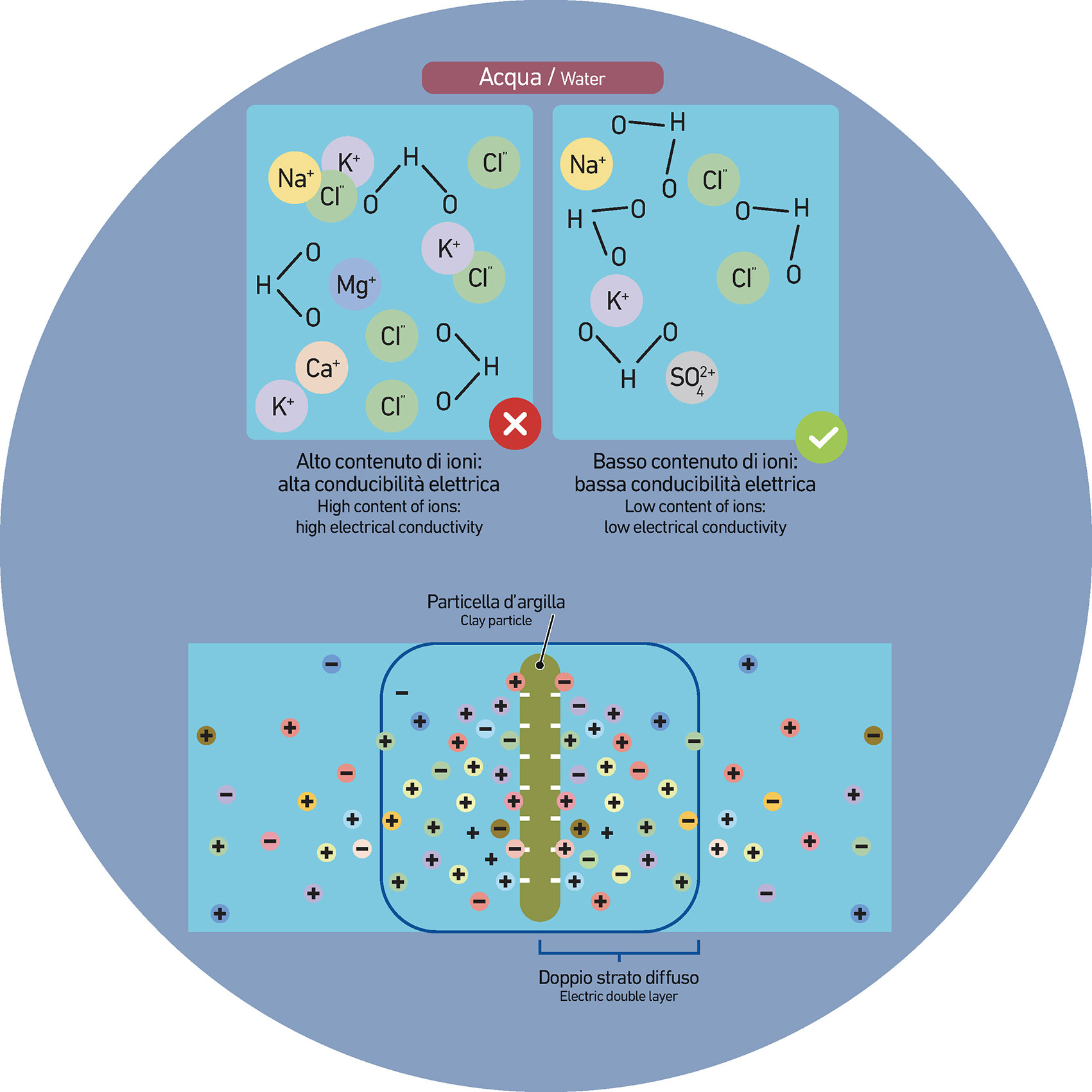
This delicate balance between electrostatic forces is highly dependent on the ionic composition of the system. Introducing electrolytes—such as common salts—adds another level of control to suspension stability. At low concentrations, electrolytes slightly compress the electrical double layer in a way that can actually enhance stability. By fine-tuning the repulsion between particles, they help maintain a dynamic equilibrium that keeps the suspension well-dispersed. However, as the electrolyte concentration increases, the double layer becomes too compressed. The repulsive forces weaken, and particles are more likely to come together, leading to flocculation or even sedimentation. At this point, the suspension may lose its stability, becoming thicker, inconsistent, or prone to settling—outcomes that can significantly affect process performance and final product quality.
C) CONCENTRATION: LIQUID TO SOLID RATIO
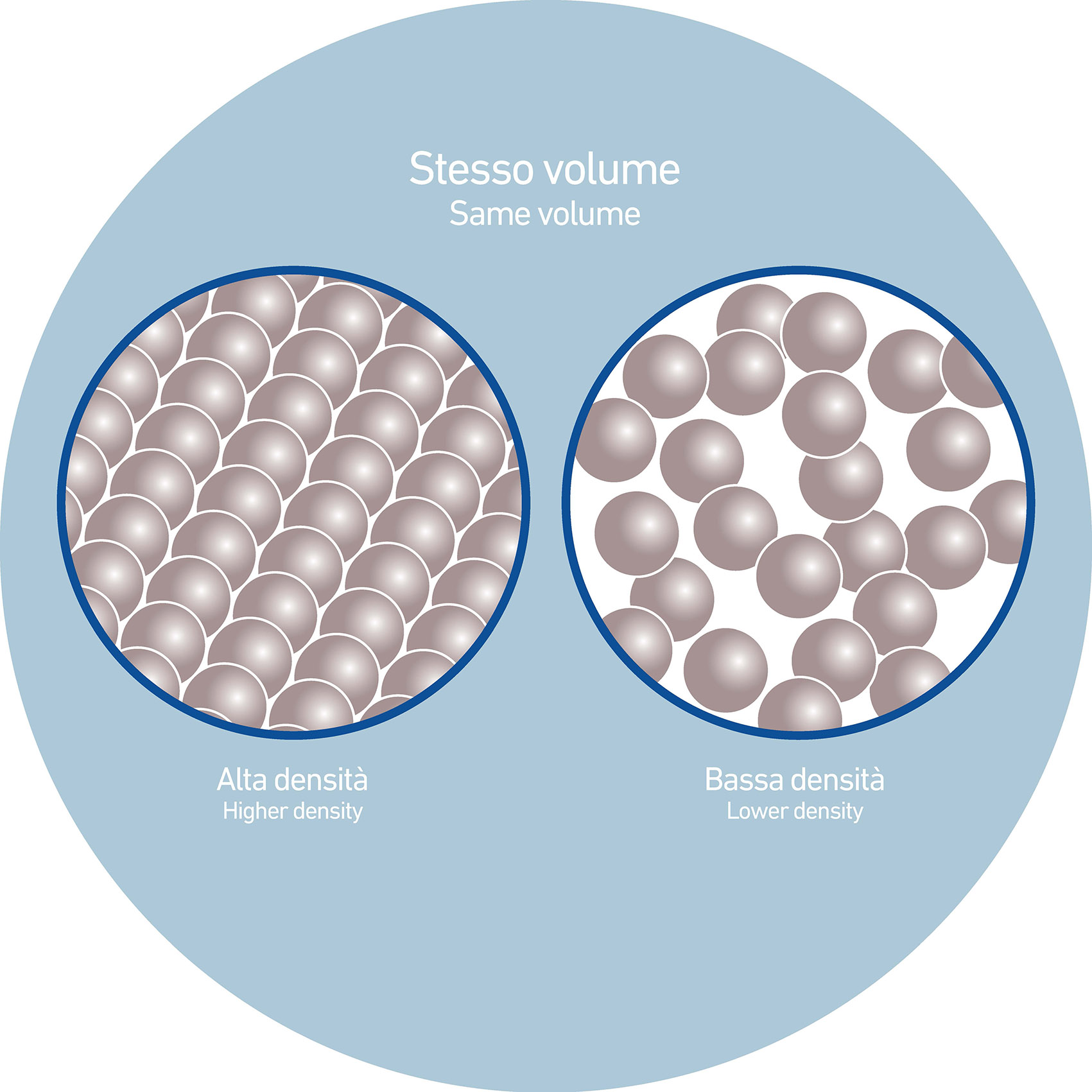
Another effective approach to improving the stability—and therefore the suspending behaviour—of a ceramic system is by adjusting the liquid-to-solid ratio. What does this mean in practice?
Put simply, the farther apart solid particles are from each other, the more freedom they have to move—and, as a result, the more likely they are to settle. Now imagine reducing the amount of water in the system: as the liquid content drops, the particles are brought closer together.
This decrease in the distance between particles leads to more friction within the narrow spaces (interstices) between them, which in turn slows down their movement. The liquid has a harder time flowing freely between closely packed particles, and this restricted mobility helps reduce sedimentation.
In short, a more concentrated suspension promotes stronger interaction between the liquid and solid phases. All other conditions being equal, this tighter packing makes the system more resistant to settling.
Of course, it’s always a matter of finding the right balance. Pushing the system too far in terms of concentration can make it so dense and viscous that it becomes impossible to process with the standard equipment used in ceramic production.
In such cases, the solution may lie in introducing a plasticizer or dispersant additive. These products can help improve the flow properties of the suspension, making it easier to work with—even when the water content is low and the stabilizing effect is strong. This way, the system remains stable without compromising processability.
3. Suspending action and type of suspensions
Depending on the type of suspension being formulated, it’s essential to choose the most suitable stabilization strategy among those discussed. This choice should always consider that suspension stability often results from a careful balance, and in many cases, a combination of approaches may be required to achieve optimal results.
A) SLIP (BARBOTTINA)
When working with ceramic slip, the most effective suspending strategy generally involves focusing on concentration, specifically the liquid-to-solid ratio. In these systems, the goal isn’t so much to use a suspending agent with thickening properties, but rather to develop an effective fluidification system.
Using a high-viscosity additive to keep solids suspended is typically not advisable here. That’s because one of the key priorities for ceramic manufacturers is minimizing the water content in the slip to reduce drying time and, as a result, lower energy costs. The best approach in this case is to maximize concentration: reducing water content while maintaining good dispersion. This achieves two things at once: a more stable, highly concentrated suspension with less water to evaporate during spray drying, and reduced sedimentation over time.
B) GLAZE SUSPENSIONS
When it comes to glaze suspensions, the choice of additive often depends on the application technology being used—such as spray guns, vela, bells, or dosing valves systems. Glazes can be applied at different stages of the production line: directly onto the body, after engobe application, or even as a final coating after grit application (in the case of dry salt finishes, for example). Depending on when and how the suspension is used in the process, different additives may be needed to adjust its rheological behaviour. Tailoring the formulation in this way ensures the glaze performs optimally, delivering consistent application results across the chosen equipment and process step.
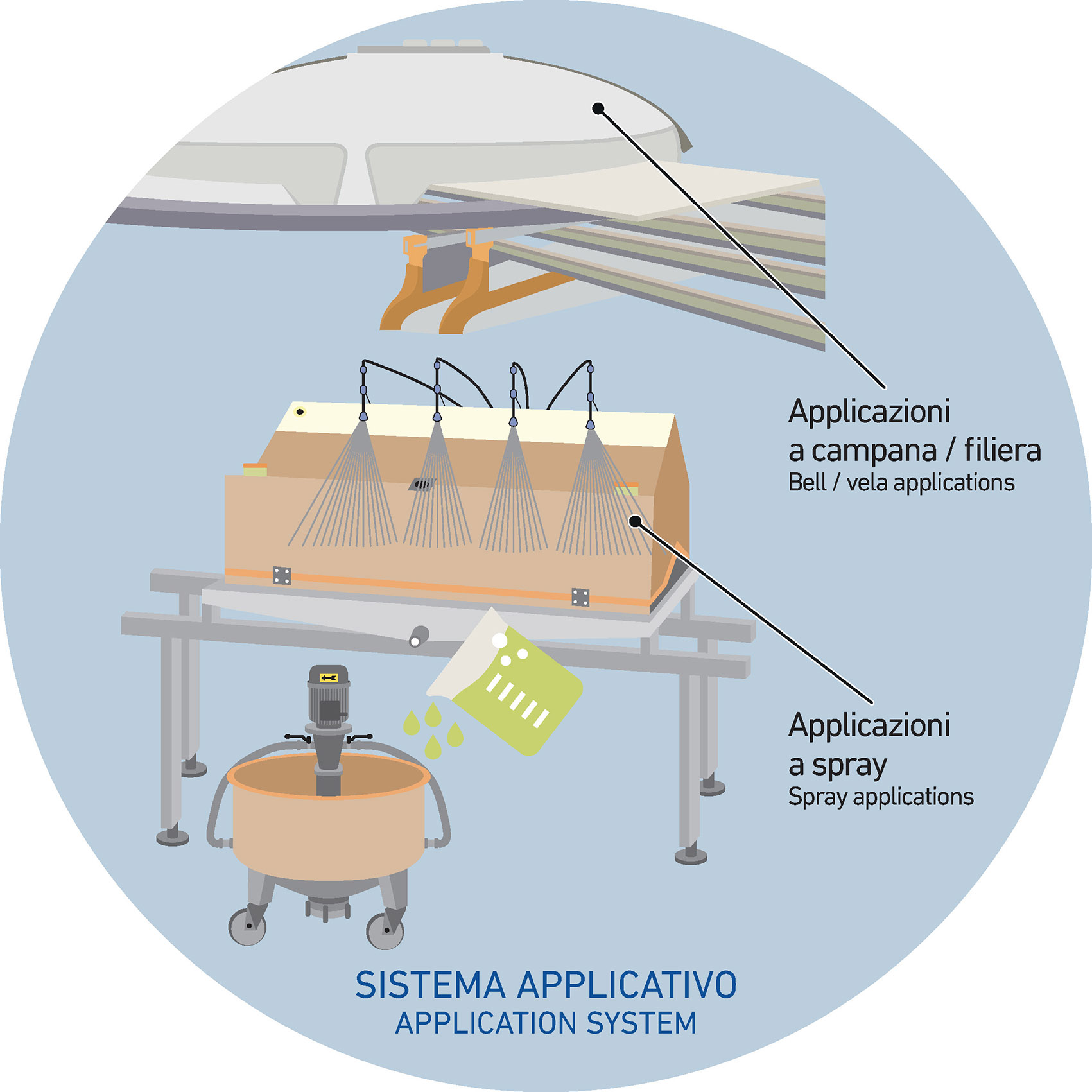
Bell applications
For bell application, glaze suspensions must be highly concentrated, which means achieving high density through a carefully optimized liquid-to-solid ratio. The goal is to maintain a cohesive, well-structured system that supports the specific requirements of this application method, while also providing sufficient suspension stability within the tanks along the glazing line.
Since this technique demands fast drying times, it’s generally more effective to control water content rather than rely on traditional suspending agents. In this context, the use of fluidizers and binders becomes more important—they help regulate application flow and ensure consistency across the surface.
To maintain good particle dispersion and prevent sedimentation, it’s essential to strike the right balance between dispersants and binders in the formulation.
Airless applications
Airless systems work with much more diluted glaze suspensions compared to bell application. The degree of dilution—and the possible need for suspending agents—depends largely on where the application occurs within the glazing line.
For example, final-stage applications (such as salt-style finishes) require very low solid content and high dilution. On the other hand, when airless systems are used upstream in the process, such as for engobe application after the dryer, the solid content must be considerably higher.
While it’s technically possible to apply glazes in this second scenario without suspending agents, their presence can often make the process smoother and more stable—especially when working with thicker, heavier formulations.
Valve applications
When using valve-based glaze application systems, careful formulation is essential. One of the biggest concerns is preventing sedimentation within the internal circuits of the machinery, which can disrupt the production process.
As a general rule, it’s important not to overload the suspension with stabilizing additives. Valve systems typically operate within a well-defined rheological window, and maintaining the right balance between suspension stability and application performance is key.
In other words, a perfectly stable suspension might still perform poorly during application—resulting in uneven or inadequate surface coverage. Conversely, a suspension that shows slight sedimentation might apply beautifully on the surface, but could compromise machine efficiency or lead to blockages over time.
For this application method, optimizing the rheology of the glaze suspension is critical. The formulation must perform well both in the tank—where it must resist settling—and in the machine, where it must flow consistently and coat the surface evenly. This often requires the precise use of rheology modifiers, tailored to the demands of both the glaze composition and the production setup.
C) GRIT SUSPENSIONS
When it comes to grit suspensions, the main strategy for achieving stability relies heavily on the use of rheology modifiers—in other words, specific functional additives. In this context, their role is absolutely essential. The reason is quite straightforward: compared to other types of ceramic suspensions, grit contains much larger particles, which makes sedimentation happen faster and more aggressively. As a result, the additives used in these formulations need to be particularly effective and robust, capable of keeping heavy particles suspended over time.
In short, ensuring proper stability in grit suspensions means choosing the right rheological additives and using them at appropriate dosages to counteract the natural tendency of the material to settle quickly.

