#41 Chemicals & seasonability: problems and suggestions
- Introduction
- Ceramic mixtures & dispersants
- Bacterial degradation
- Change in the evaporation & drainage times
- Solubility of surfactants
1. Introduction
In the ceramic industry, the use of additives plays a crucial role in ensuring that the production process meets all the standards and in achieving products with high technical and aesthetic performance.
Chemicals are also called vehicles because they convey specific properties within the suspensions (or more generally, mixtures) to which they are added.
However, the action of additives can sometimes be compromised, less effective, or otherwise altered.
There are several reasons that can affect the performance of an additive, among which seasonality certainly plays a significant role.
What does that mean?
In simple words, it can be stated that a ceramic semi-finished product (such as a glaze suspension, engobe, grit suspension, etc.) containing one or more additives may show different behaviour depending on the season and, to some extent, the temperature.
From another perspective, one could also say that when external conditions change, the additive might require some adjustment to maintain the already defined standards.
Given the wide range of possible scenarios, this discussion addresses a few cases aimed at highlighting the existence of the phenomenon and emphasizing the complexity of the balances that must be taken into account whenever operating along ceramic production lines.
2. Ceramic mixtures & dispersants
One of the most frequent requests received by chemical laboratories with the arrival of the hot season (generally from June to September) concerns the revision of deflocculant formulations. These agents seem to have partially lost their effectiveness, no longer being able to prevent excessive increases in the viscosity of the slip. In some cases, this increase can even lead to complete jelly phenomena within the system.
What happens? Is the problem due to an improperly formulated additive, or have the parameters and conditions of the suspension simply changed?
As is well known, the main components of slip are a mixture of raw materials (clays, feldspars, etc.), chemicals, and water. For industrial productivity reason, ceramic producers reduce the water content as much as possible to speed up the evaporation process and, consequently, reduce energy costs.
The formulation of the chemicals involved must, therefore, take this necessity into account to make it feasible.
Nevertheless, even though the water content is kept to the minimum (or perhaps because the content is deliberately low), water plays a crucial role in the critical issues that typically arise towards the end of summer.
Why?
The process waters used in ceramics, and in particular within the grinding process (preparatory to the production of barbottina) have several origins. Among these, groundwater is the most important source of water.
However, groundwater is subject to all seasonal variations throughout the year. In particular, during the dry seasons, due to the scarcity of rains, the level of the most superficial aquifers changes and therefore it also changes the content of salts. In other words, the water table rises or falls – often significantly – due to rainfall or prolonged drought.
These rises or falls in the levels have a very important impact on the content of dissolved salts and therefore also on the electrical conductivity values of the waters which affect the rheological parameters of the barbottina, often by modifying them and thus making the action of dispersants less effective.
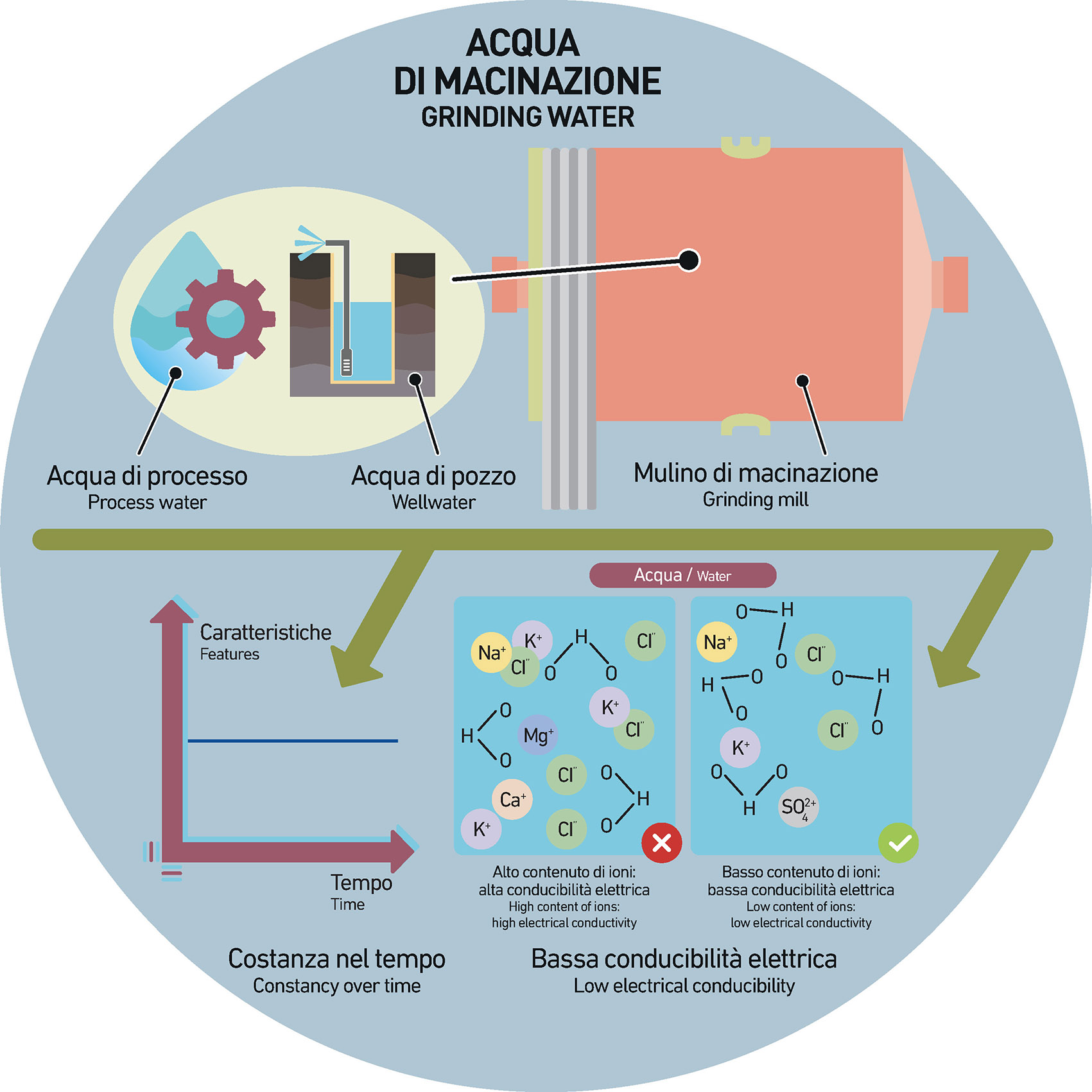
In simple words: groundwaters extracted during the winter or spring season are marked by quite different features from the waters that come from the same aquifer just before the beginning of autumn.
The decrease in water level causes an increase in the percentage of salts, which results in a significant increase in the content of ions that in turn affects the electric double layer of particles that are in suspension within the barbottina.
The grinding process, however, involved not only well water (more controlled) but also waste waters that come from several departments of the production line. These waters are often rich in contaminants such as salts and bacteria and, therefore they may worsen the scenario.
What does it mean?
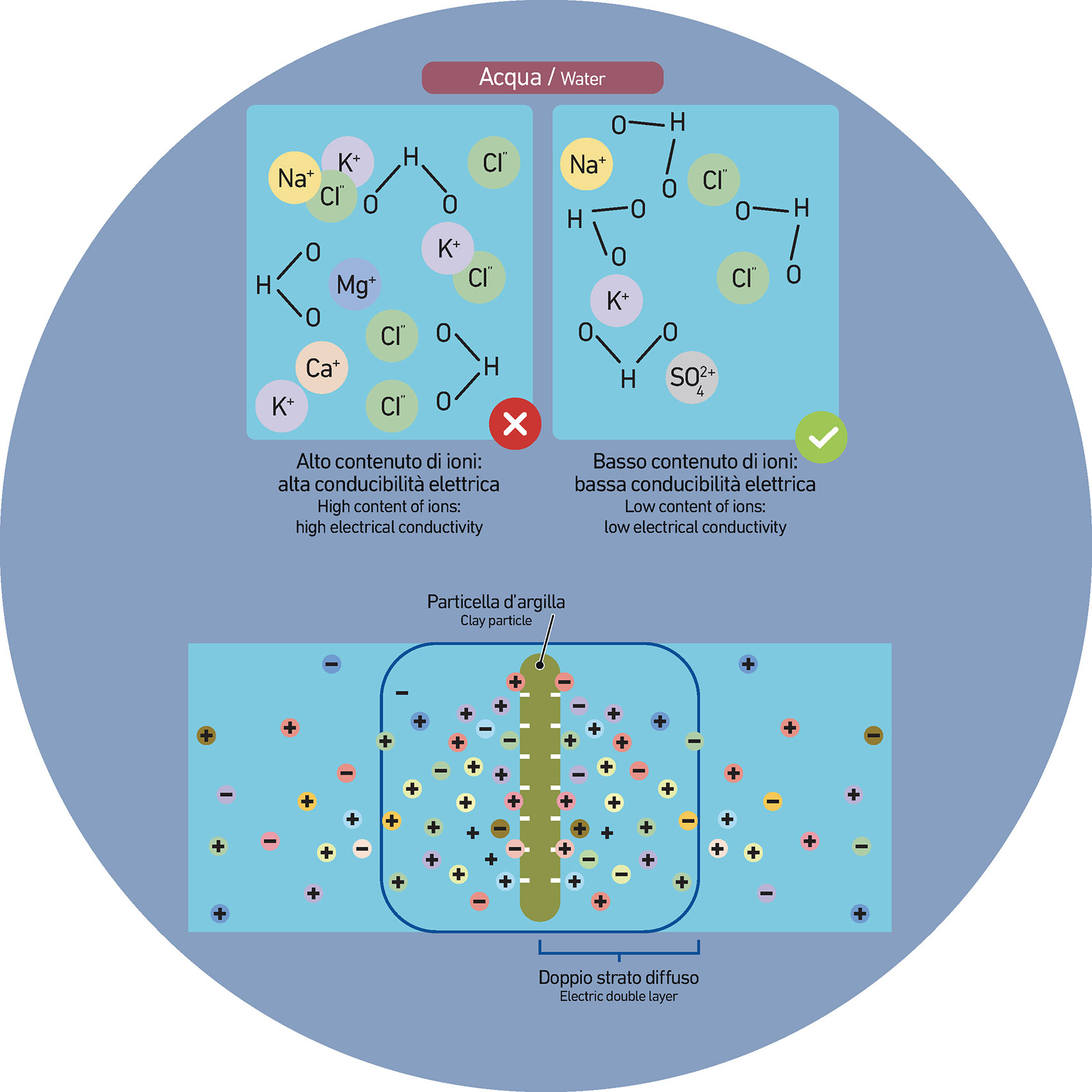
In general, it is important to keep constant the composition and the quality of water over time so as to avoid possible fluctuations in the grinding parameters which would force companies to continuously adapt and modify the production parameters. For this reason, the water must be constantly checked and controlled, especially in terms of pH and electrical conductivity which essentially indicates the amounts of ions (cations and anions).
A significant percentage of multivalent anions and cations (such as calcium 2+ or magnesium 2+) can interfere with the fluidization process by reducing the thickness of the diffuse layer on the surface of clay micelles, that is the distribution and thickness of anions and cations on their surface.
This reduction results in a general increase of the viscosity of the slurry deriving from the presence of electrostatic attraction phenomena. We could say, simplistically, that the solid parts of the suspension interact by increasing the viscosity.
The above-mentioned criticality arises not only because the conditions of the groundwater have changed due to seasonality but also because of the low amount of water involved within the ceramic suspension. A higher water content could certainly mitigate the problem as it would allow to operate with a wider range of options.
What can be done to better manage these scenarios?
The most immediate and simple answer, but hardly achievable because of the industrial productivity, consist in increasing the water within the system with a consequent decrease in the percentage of ionic content but with a simultaneous increase in energy consumption necessary for the subsequent evaporation process. The second-best alternative, more complex but often inevitable, is to work on the formulation of the dispersant agent, which consists largely in revising the percentages of the different components of the “recipe” until the physical viscosity parameters are reached, allowing a proper application of the barbottina.
This confirms that the same dispersant, at the same concentration, can give rise to very different performances according to the ionic content inside the process waters used for grinding ceramic mixtures.
3. Bacterial degradation
Seasonality, and more generally temperature, can also affect the possible appearance of bacterial proliferation within semi-finished products. Why do we store food inside our fridges, for example? Because bacteria usually find a perfect habitat for their reproduction when temperatures are approximately around 35/37°C: temperature is one of the elements that most affect bacterial growth.
More specifically, bacteria grow best within a temperature range of 20 °C to 40 °C, with an optimal growth temperature for most bacteria of around 37 °C. At lower temperatures, bacterial growth slows down or stops completely. There is more: the growth rate of the bacterial proliferation increases with temperature until the so-called thermal death temperature is reached, which for most bacteria is about 60 °C.
Before being used, semi-finished ceramic products (glaze or grits suspensions, barbottine, etc.) often rest for longer or shorter periods inside storage tanks, greatly increasing their temperature during the hottest periods and somehow facilitating the reproduction of bacteria that are naturally present in the environment.
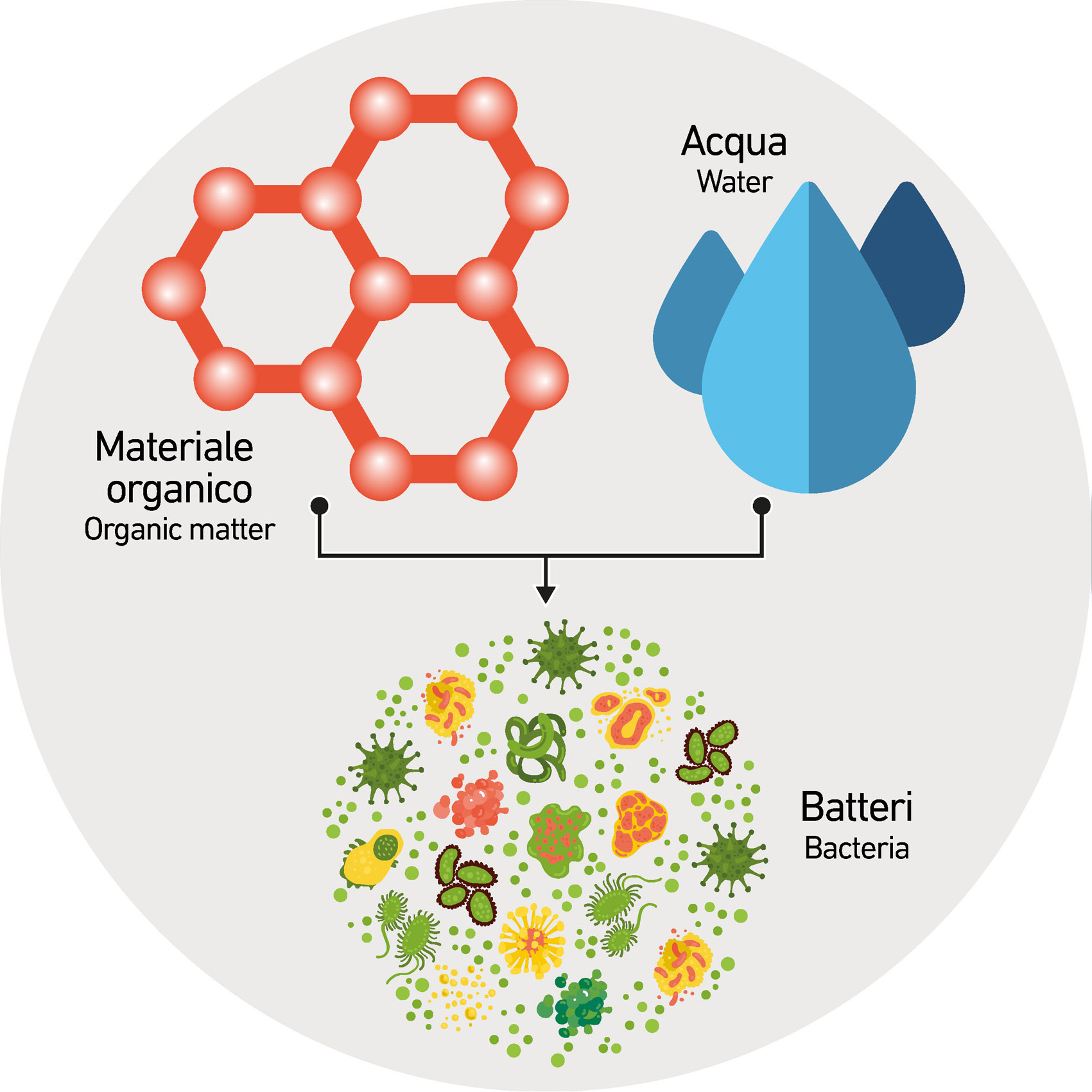
The most favorable situation for bacteria is the simultaneous presence of water and organic material that are their main source of nourishment: the higher the temperature of the semi-finished product and the surrounding environment, the faster (and therefore more impacting) will be the bacterial proliferation and degradation of the suspension.
In all respects, bacterial proliferation produces a degradation of suspensions and in particular of chemicals, weakening their action and therefore losing the correct rheology parameters. A process that inevitably affects the proper development of the process or that can cause important defects in the finished product (boiling, melting, etc.).
What are the most common scenarios?
- Significant variation in the rheological parameters of suspensions
- Violent and rapid sedimentation phenomena inside the semi-finished product
- Alteration of drying and drainage times during the application of glazes and engobes
- Improper laying
- Application defects such as the appearance of holes
- Dusting phenomena of the glaze
To briefly summarise:
in the summer or in any warmer period, bacteria greatly increase their activity by “eating” chemicals, producing metabolites, multiplying disproportionately and thus generating a negative effect on the rheological properties of the semi-finished product that loses its stability and therefore its correct performance.
In addition to the inevitable storage times that certainly promote the appearance of the problem, bacteria can find a very comfortable habitat even inside the tanks, of the tubes, pipes and all those points in the production plant where a stagnation of organic material and water content can occur. It follows that whenever a suspension free of bacterial contamination comes into contact with a source of contamination (such as those described above) it is itself contaminated.
How to manage this problem?
In these cases, the most sensible action would be a preventive approach by careful monitoring the working environment (both storage and application areas) to check for the presence of bacterial colonies and carry out an appropriate sanitization.
In the case of existing proliferation, two actions are normally taken:
- Elimination of bacteria by using appropriate biocides and preservatives in a correct amount
- After careful laboratory analysis, restoration of the rheological characteristics of the suspension through the use of the most appropriate additives for the purpose
To prevent further contamination or re-contamination of the suspensions, it is advisable to carry out periodic sanitizations of the plant (pipes, tanks, etc.) in order to minimize sources of contamination and prevent possible future re-contamination.
4. Change in the evaporation & drainage times
This kind of problem generally applies to all ceramic suspensions but what is it? The watery ceramic suspension, whether it is an engobe, glaze or grit suspension, is discharged on the hot ceramic support which, thanks to its high temperature, facilitates and acts on the evaporation process.
Normally, when all other parameters are equal, the temperature of the tile may vary according to seasonality: during the hottest months, the outside temperature increases. This increase, even if of few or very few degrees, can affect and modify the performance of applications along the glazing line.
It is also possible to say that in this respect some areas of the line are more susceptible than others: ideally the positions which are located further from the dryer could have very different temperatures (with the same parameters and according to the season).
What does it mean?
Once the ceramic material has left the kiln, it keeps on going along the glazing line where it receives, layer by layer, the different applications: ideally, it is important to make sure that the temperature of the support remains unchanged, regardless it is summer or winter.
After that, the tile gradually begins to cool and this cooling will happen more quickly as the temperature difference between the tile and the surrounding environment is greater.
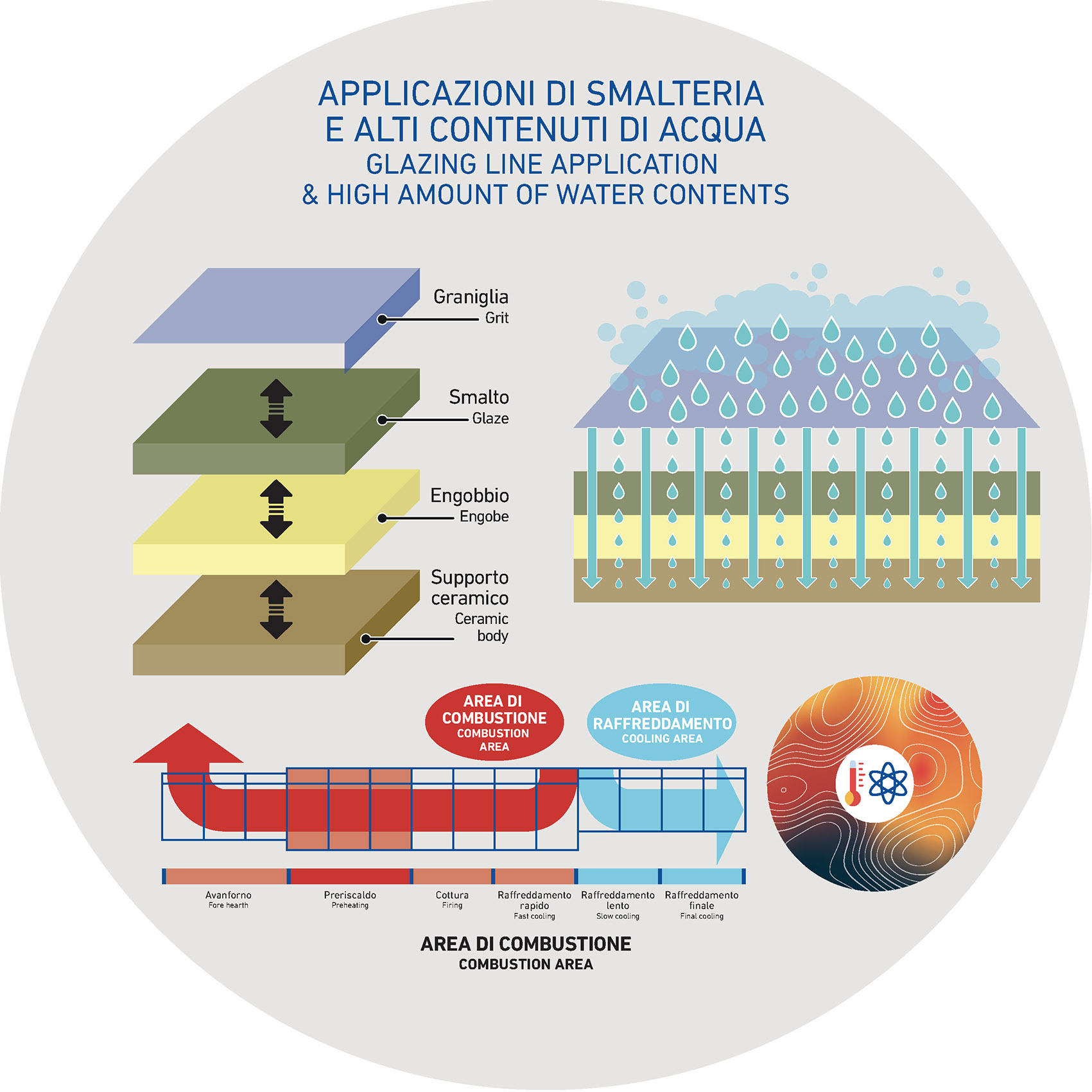
Winter temperatures will cause a greater difference in temperature between the environment and ceramic substrate compared to the summer situation. This will cause a higher cooling rate of the media during the winter season compared to summer and, as a last result, you can see a variation in drying times of various applications (faster in summer, slower in winter).
A trivial but useful example: think about a tile that, after the dryer, is about 110°C.
Imagine carrying this support along the glazing line in two different scenarios: in summer and winter.
The first application (for example an engobe), with the same line parameters, may not show differences compared to the two different situations (summer and winter) because the tile keeps in both cases the same temperature.
With the subsequent applications, however, the conditions can change significantly as the tile, depending on seasonality, may cool with different times (faster in winter, less quickly in summer), unevenly affecting the drying and draining times of the applied suspension.
The water evaporation, of course, will also be faster in summer and slower in winter due to different temperatures of the environment.
What can happen?
In general, drying times in the winter can be longer (under the same conditions) and the duration of drying and drainage times is in some way related to the risk of defects appearing on the support.
As a rule, the longer duration of the evaporation and drainage process – unless specifically sought – can help a greater number of problems that do not allow a perfect discharge of the suspension on the support. Some potential problems are listed below as examples:
- During the application of glazes and engobes, differences in application can be observed between summer and winter;
- During the digital printing decoration, the quality of the application may be affected due to different ink absorption rates by the ceramic support, or (in winter) water condensation may appear under the digital printer bars;
- During the firing cycle, potential problems may arise in the preheating phase of the kiln due to improper drying of the tiles before entering the kiln
Even in this case – speaking of a production project that is continuous throughout the year or that takes place at very different times of the year – chemicals are required to prevent or at least to reduce to a minimum the differences in applications between summer and winter. This is important to avoid strong actions on other parameters, keeping them unchanged.
To ensure the smallest difference in drying times between summer and winter, for example, it should be necessary to optimize the amount of water, reducing it as much as possible (according to the type of application).
Why?
A lower water content is a synonym for less drying time, reducing therefore the problems that we have shortly described. In any case, the rheological characteristics of engobes and glaze suspensions must be balanced to ensure perfect application despite the reduction in water content.
5. Solubility of surfactants
An important premise: the category of surfactants is huge and extremely various. They are numerous and marked by very different properties. They certainly do not have an universal use but must be studied and chosen according to the mixture to which they are applied.
Surfactants are strongly influenced by temperature and indirectly by seasonality. Especially when it comes to solubility.
Variations of 15° or 20 °C, as it happens between summer and winter, may affect their performance.
The change in the solubility and critical micellar concentration (CMC) of the surfactant inevitably affects its action on the surface tension (its main function), in some cases no longer meeting the standards.
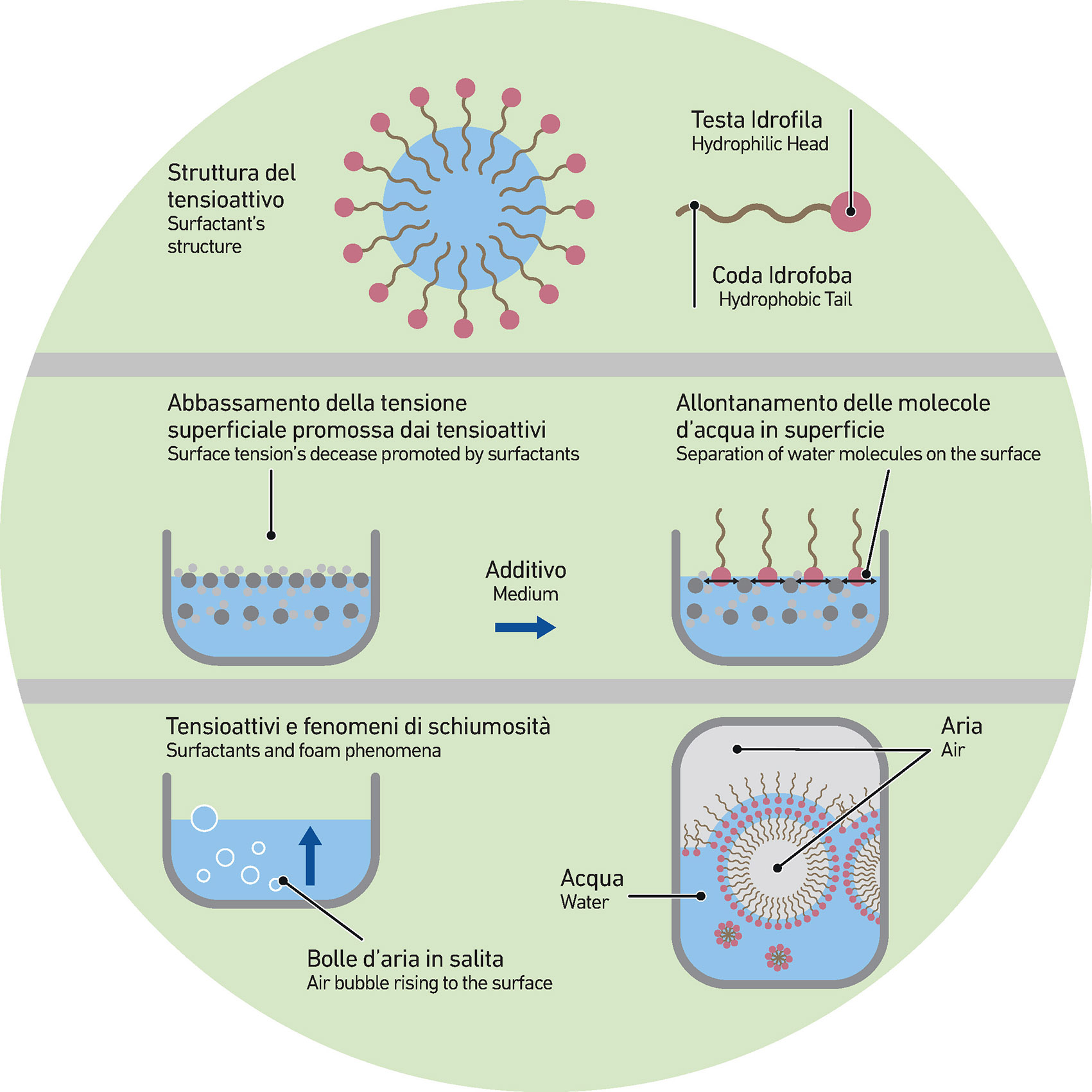
A clear example.
Let’s consider a solution/suspension consisting of 99.9% water and 0.1% surfactant. It can happen that at 5°C the system is completely clear and marked by foaming phenomena. The same system at 25°C could be opalescent and foam-free. This is because some products (surfactants) are characterized by a solubility that changes (in this case decreases) according to the temperature.
Conversely, there are also cases where the behavior is exactly the opposite: with the increase of the temperature of the system, the solubility of surfactants increases.
In other words, a given surfactant leads to excellent performance when used within a certain temperature range while if you change the temperature, it might be unsuitable for the same kind of process. This tells us, once again, that the surfactant must be tested time after time according to its use, checking its performance.
What should be done when a surfactant seems to be ineffective? The most frequent issues due to the difference in temperatures between summer and winter are usually about to the greater difficulty of dispersing these chemicals within the suspensions. During the winter periods it is good to keep in mind that increasing the mixing times can solve the criticality.
If these small steps do not help, there are two possible solutions:
- Revise/replace product or recheck dosage
- Use the product in combination with other additives (to be checked and tested from time to time) so as to obtain a better blend based on the problem that must be solved (Example: add a defoamer if foaming occurs)

