#50 Reverse osmosis: challenges & solutions in water treatments
- Osmosis and reverse osmosis: an overview
- Benefits of reverse osmosis in water treatment
- The reverse osmosis process: challenges and solutions:
- a. Antiscalants
- b. Reducing agents
- c. Coagulants
1. Osmosis and reverse osmosis: an overview
Water is one of the most widely used natural resources—if not the most used—across various industrial sectors and applications. Process water plays a key role in enabling and supporting different stages of production. However not all water is the same. Depending on its source—whether it comes from recycling systems, groundwater, or municipal supply—its characteristics can vary significantly, making it more or less suitable for specific industrial uses.
In many cases, water quality is a critical prerequisite for proper processing. For this reason, several technologies are available to purify water effectively and safely. Among the most common and widely adopted methods is reverse osmosis.
OSMOSIS
To fully understand reverse osmosis, it’s important to start with the basic concept of osmosis.
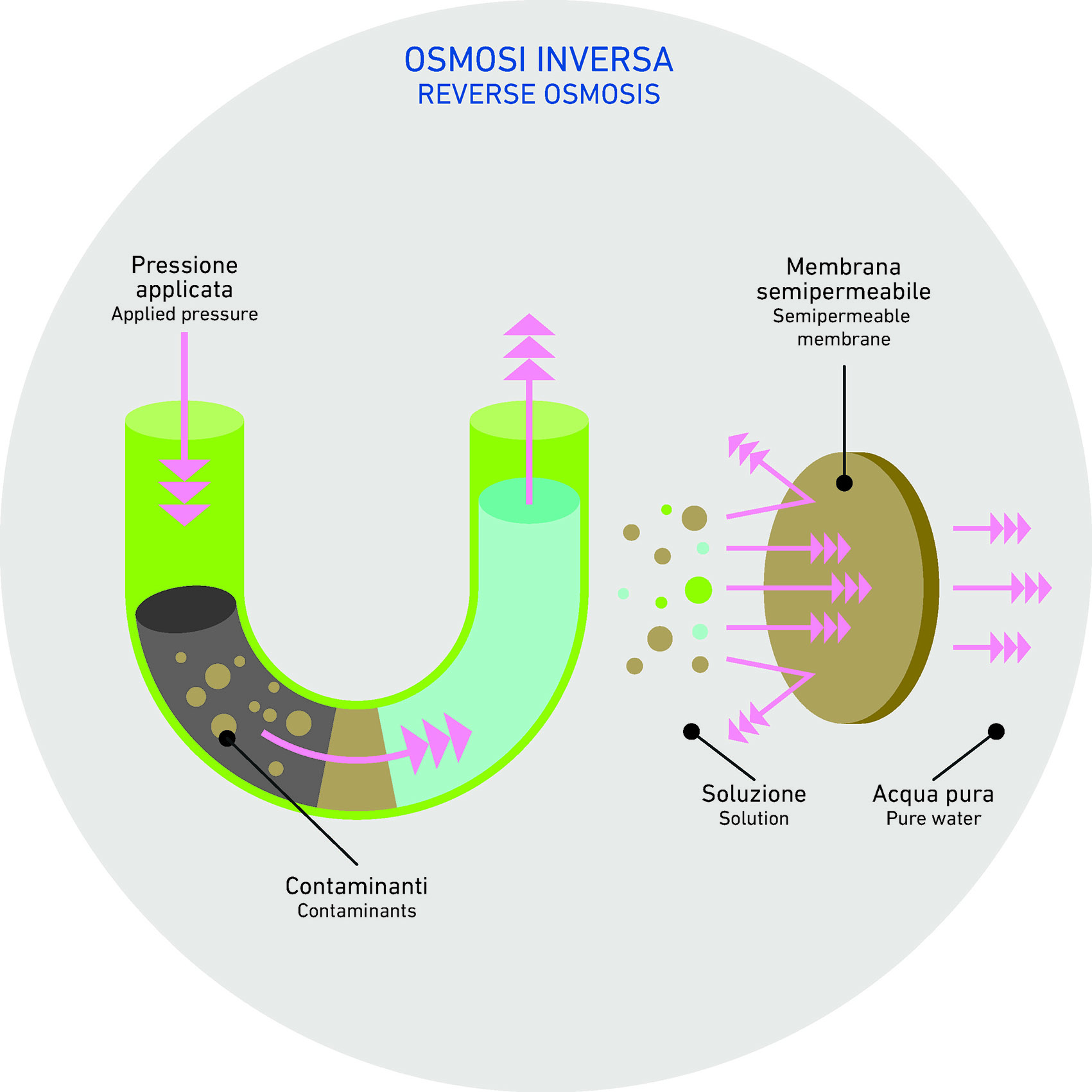
Osmosis is a natural phenomenon where a solvent—typically water—moves across a semipermeable membrane that separates two solutions. This movement is usually driven by a concentration gradient: the solvent flows from the less concentrated solution to the more concentrated one.
However, osmosis can also occur due to differences in electric potential (electro-osmosis) or temperature (thermo-osmosis).
The goal of this process is to equalize the concentration of dissolved solids on both sides of the membrane. This movement continues until equilibrium is reached, and the driving force behind it is known as osmotic pressure.
REVERSE OSMOSIS
As the name implies, reverse osmosis is the opposite of the natural osmosis process—and it requires external force to occur.
Reverse osmosis is a separation technique that uses a semipermeable membrane and applies external pressure to reverse the natural osmotic flow. In this case, the solvent (usually water) is forced to move from the more concentrated solution toward the less concentrated one.
This is only possible by applying a pressure greater than the natural osmotic pressure. In practical terms, water containing a high concentration of dissolved solids is pushed through the membrane, leaving behind impurities and allowing purified water to pass through.
As a result, the process yields two distinct streams: a concentrated waste stream containing all the dissolved solids, and a purified stream of demineralized water.
Reverse osmosis, therefore, is a non-spontaneous process that requires mechanical energy to overcome osmotic pressure. The semipermeable membrane plays a critical role: it blocks the passage of solutes while allowing only the solvent to pass, making it an effective method for producing clean water from contaminated or mineral-rich sources.
In recent years, this system has gained widespread attention, largely due to the growing popularity of home reverse osmosis filters, which have made the technology more accessible to the public.
Beyond domestic use, this seemingly simple water treatment process has also seen significant adoption in industrial and agricultural sectors, where water quality is a critical factor in operational efficiency and product quality.
2. Benefits of reverse osmosis in water treatment
Reverse osmosis is considered the most advanced water filtration technique—not simply because it relies on pore size to block particles, but because it also leverages the chemical affinity between substances and the membrane. This allows the selective passage of hydrophilic (or “water-like”) molecules, such as short-chain alcohols, while rejecting others.
In simpler terms, reverse osmosis effectively removes a wide range of impurities from water, including salts, pathogens, synthetic organic compounds, pesticides, and most contaminants.
The result is high-purity water with low mineral content and no harmful substances. To illustrate its efficiency, reverse osmosis membranes can filter particles as small as 0.0001 microns.
Additionally, reverse osmosis systems are environmentally friendly: they consume relatively low energy and do not release pollutants into the environment. Beyond desalination and water recovery, these systems also allow for the recovery and reuse of valuable minerals—supporting a circular economy approach.
3. The reverse osmosis process: challenges and solutions
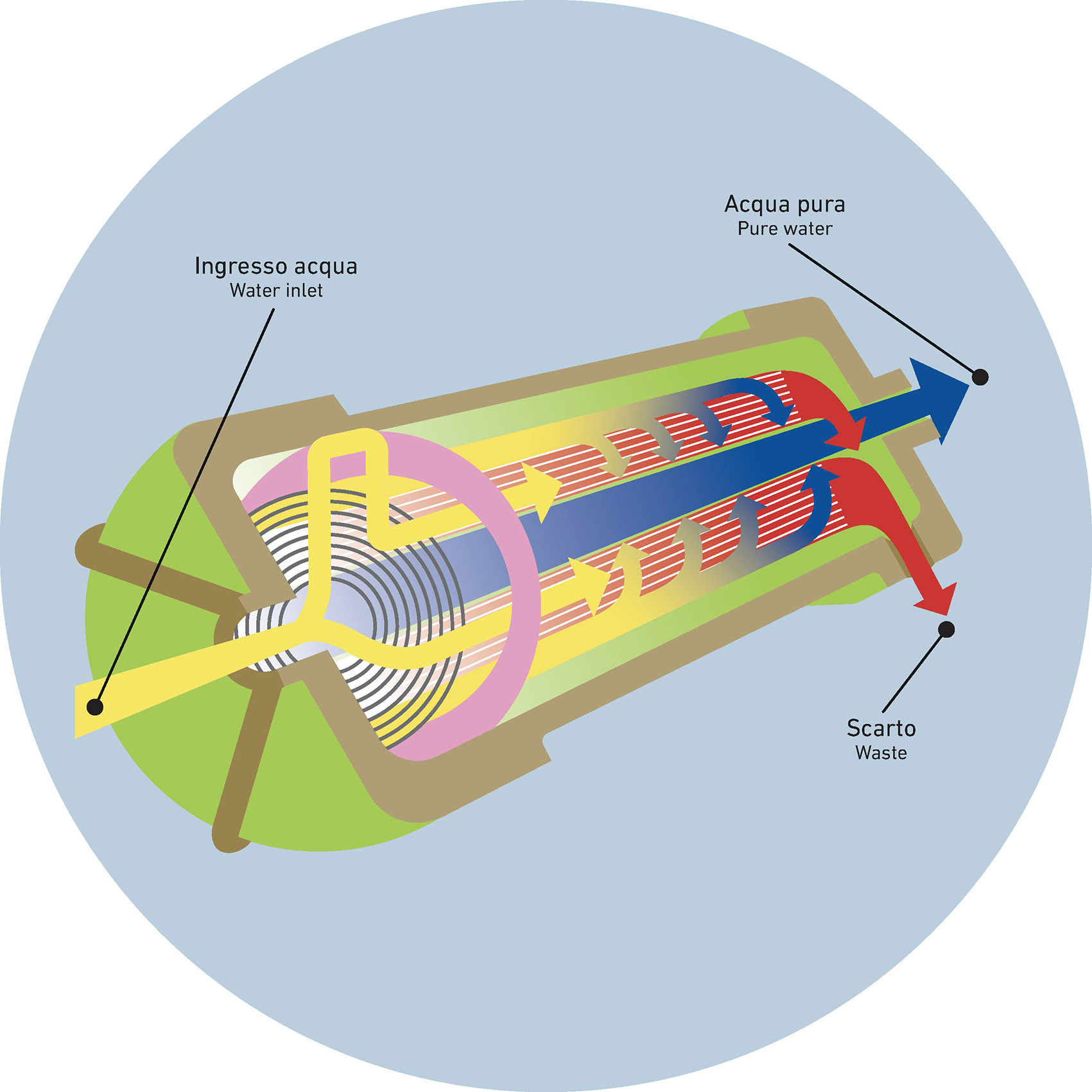
Without going into technical detail, the process begins with the intake of raw water. This is typically done using a pumping system that directs the water to the treatment unit. The quality of the incoming water, the plant’s operating parameters, and concentration polarization all have a direct impact on system performance.
If not properly monitored and managed, these factors can negatively affect the reverse osmosis membrane—leading to fouling, salt scaling, or, in more severe cases, membrane degradation.
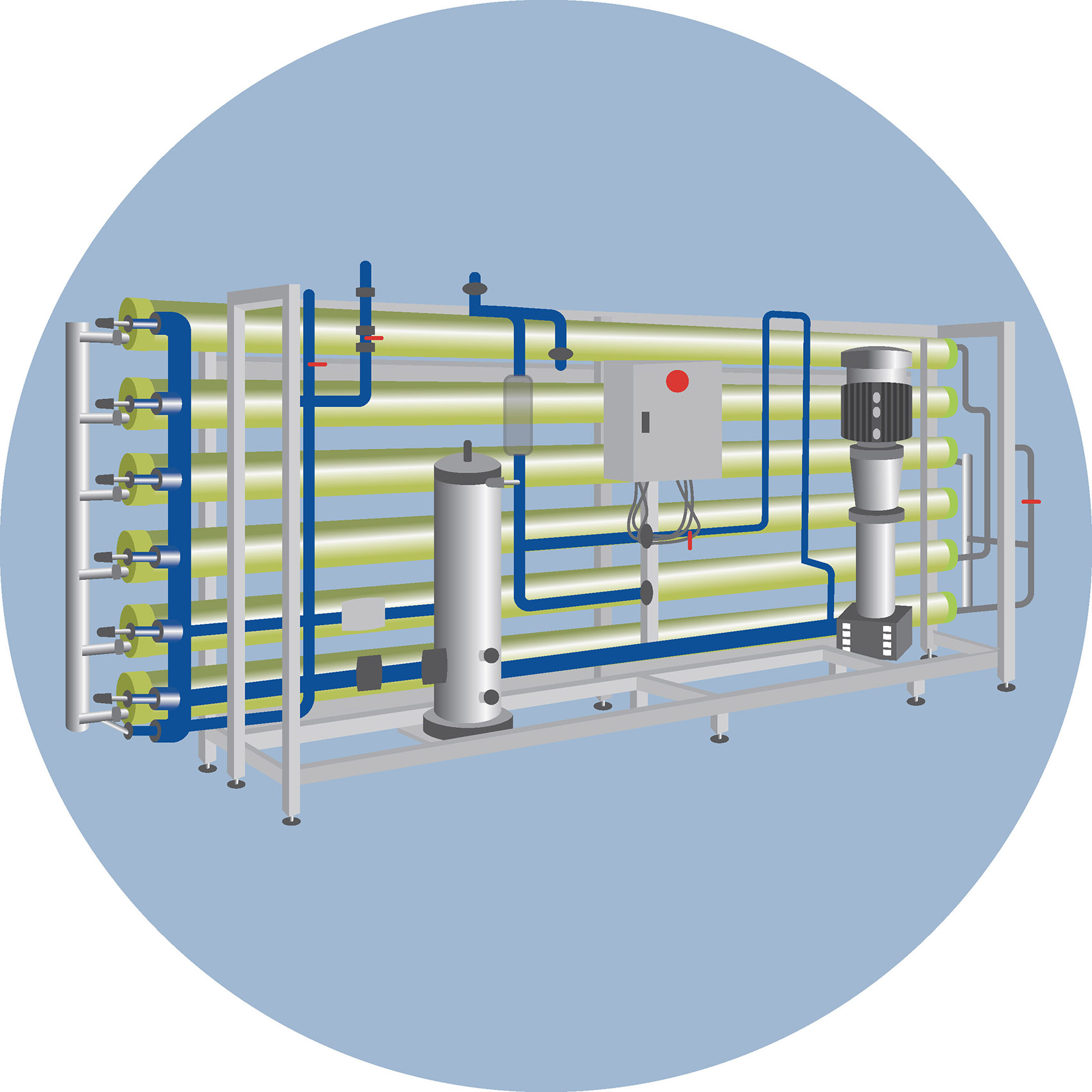
Reverse osmosis membranes are usually made of polyamide, a material selected for its high-water permeability and resistance to dissolved impurities. It effectively blocks salts and small molecules that cannot be filtered by simpler systems. (Another common example of a semipermeable membrane is the one used in dialysis.)
These issues, however, can be effectively managed—and often entirely prevented—through proper pretreatment of the water, using specific additives that improve its quality and prepare it for the reverse osmosis process.
In addition to undergoing various physical treatments, water often requires chemical conditioning using specialty additives such as antiscalants, reducing agents, or coagulants. These substances help optimize water properties and protect the membrane, ensuring stable and efficient system performance.
A. ANTISCALANTS
Antiscalants are chemical additives designed to prevent the buildup of slightly soluble salts in water.
They work by inhibiting the precipitation of these salts, thereby avoiding sediment formation and helping to maintain optimal system performance. For this reason, they are essential components in most water treatment systems.
One of their primary applications is in reverse osmosis systems, where they play a key role in preventing membrane scaling. However, antiscalants are also widely used in other systems prone to scaling issues—such as cooling towers—where the progressive accumulation of particles on surfaces can lead to performance losses or even complete blockage. Scaling is an undesirable phenomenon that can occur during nanofiltration and reverse osmosis, and antiscalants are critical in minimizing its impact and extending the operational life of the system.
B. REDUCING AGENTS
In chemistry, a reducing agent is a substance that donates electrons to another substance. During a redox (reduction-oxidation) reaction, the reducing agent can be identified by the increase in its oxidation number, which reflects the loss of electrons.
The process of reduction refers to the action of the reducing agent on another substance. The substance that receives the electrons is said to be reduced.
In water treatment, reducing agents are often used to neutralize oxidizing substances such as chlorine or oxygen, which could damage reverse osmosis membranes or interfere with other chemical processes. By donating electrons, these agents help stabilize the water chemistry and protect sensitive components in the system.
C. COAGULANTS
Among the various chemical additives commonly used in water pretreatment, coagulants play a key role in removing suspended particles and disinfectant by-products. Ultimately, they contribute to reducing the biological load in the water.
In chemistry, coagulation refers to a process in which a liquid or colloidal suspension is irreversibly transformed into a semisolid state, triggered by chemical agents (such as added electrolytes) or physical factors (like temperature changes or solvent evaporation).

