#48 Complexing agents & ceramic production: what they are and how they work
- Introduction
- What are complexing agents?
- Complexing agents in the ceramics industry
- Phosphonates: an example of a complexing agent
1. Introduction
Who hasn’t played Pac-Man at least once?
In the iconic video game, players guide a little yellow ball-shaped character through a maze, trying to eat all the dots while avoiding four chasing ghosts. If a ghost catches Pac-Man, you lose a life.
Interestingly, complexing agents—used in many fields, including ceramics—work in a similar way.
In simple terms, they “capture” multivalent cations to stop them from interfering with ceramic processes.
MULTIVALENT CATIONS
Multivalent cations are positively charged ions that carry more than one positive charge. This means they’ve lost multiple electrons. Common examples include:
Calcium (Ca²⁺): +2 charge
Magnesium (Mg²⁺): +2 charge
Iron (Fe²⁺ or Fe³⁺): +2 or +3 charge
Aluminum (Al³⁺): +3 charge
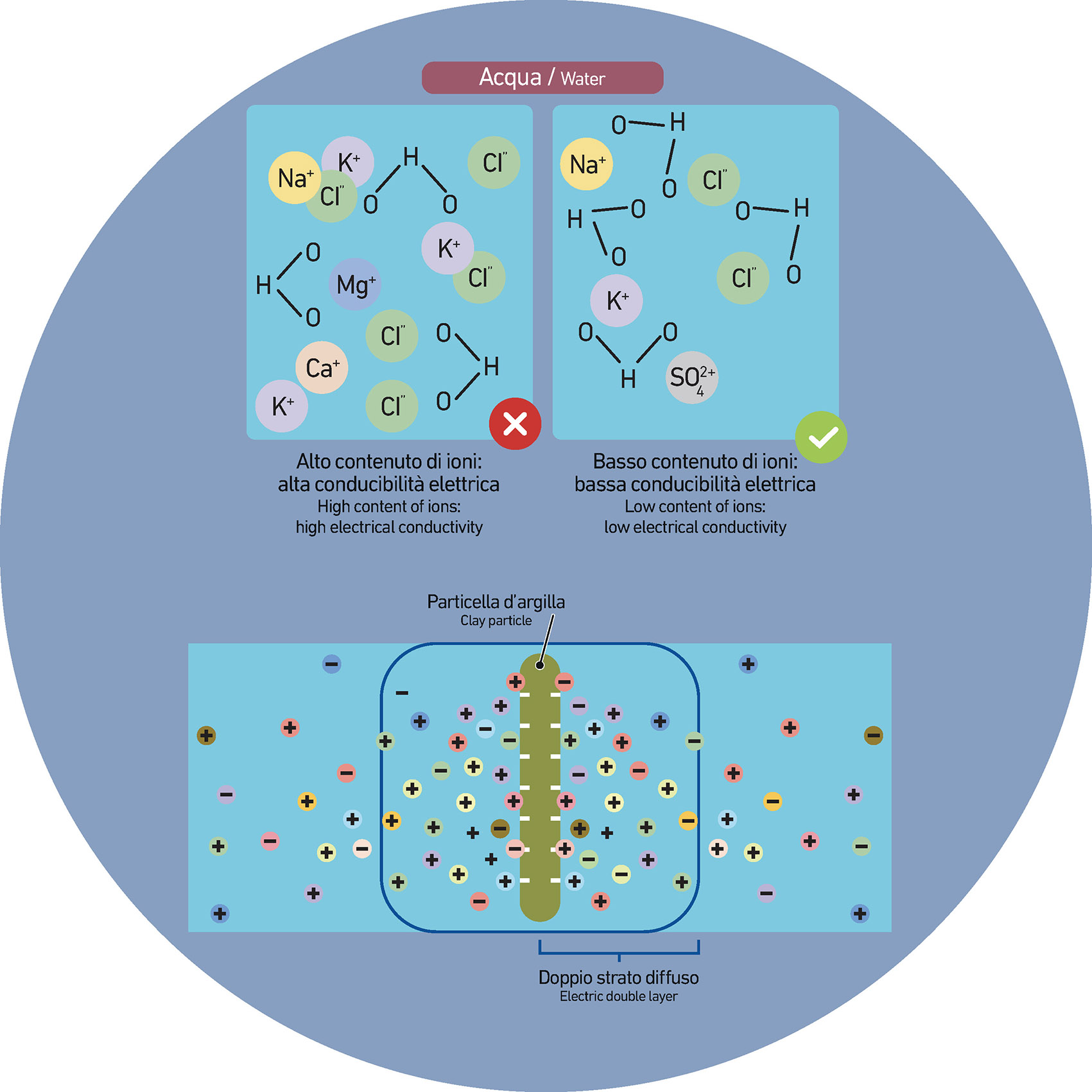
These ions play a key role in many chemical and biological reactions, including deflocculation and the stabilization of suspensions—particularly in materials science and ceramics. Their presence can significantly affect the physical and chemical behaviour of these systems.
2. What are complexing agents?
Complexing agents are essential players in one of the key mechanisms behind deflocculation: complexation. But how do they actually work?
Also known as chelating agents, complexing agents are chemicals that bond with specific ions—like calcium, magnesium, iron, or copper—creating stable compounds. This makes the metal ions less reactive and prevents them from precipitating out of solution. These agents are used across a range of industries, from cleaning products to water treatment to healthcare.
HOW THEY WORK
Complexing agents have chemical groups that can donate electrons and form coordinated bonds with metal ions, creating stable structures called coordination complexes. Once a metal ion is “captured” in this way, it becomes chemically inert and less likely to interact with other substances.
In ceramic applications
In ceramics, complexing agents are typically molecules containing negatively charged functional groups—such as oxygen or nitrogen atoms. These groups attract multivalent cations (like calcium and magnesium) found in hard water and raw materials stored in tanks, before the atomization process.
By neutralizing multivalent cations, complexing agents promote the presence of monovalent ions, triggering cation exchange. This exchange increases the distance between particles, creating a deflocculating effect and lowering the viscosity of the fluid. As a result, it’s possible to increase the solids content of a suspension (or slurry), while also reducing water and energy usage.
KEY PROPERTIES OF COMPLEXING AGENTS
Complexing agents stand out from other deflocculants due to several characteristics:
- High water solubility and versatility, making them suitable for various applications
- They don’t change chemically before bonding with multivalent cations
- Like Pac-Man eating multiple ghosts, one molecule can chelate multiple ions like calcium or magnesium
- Bonding mechanism: they can surround a metal ion or link two ions together
3. Complexing agents in the ceramics industry
To understand the role of complexing agents in ceramics, let’s go back to basics: what’s the connection between these agents and deflocculation?
Ceramic suspensions—commonly referred to as slurries—can sometimes display a thixotropic behaviour.
Thixotropy is a property of certain fluids and gels that causes their viscosity to decrease when agitated or stirred, and to increase again when left at rest. In other words, the more you stir, the more fluid they become—until they return to a gel-like state when undisturbed.
While this behaviour is interesting, it’s not always desirable in ceramics.
When clay micelles are suspended in water—especially in limited quantities—electrostatic attraction between the clay layers can cause a sharp rise in viscosity, and the suspension may even solidify into a gel over time. For the suspension to be usable, it must regain a proper viscosity level, returning to a fluid state suitable for processing.
METHOD S OF DEFLOCCULATION
There are several ways to restore fluidity and reduce viscosity in ceramic suspensions:
- Adding monovalent cations (like sodium), which replace stronger multivalent ions through cation exchange, lowering the system’s viscosity
- Using polymeric dispersants, which have long, inactive chains that prevent particle interaction and keep them separate, reducing viscosity
- Applying complexing agents, which neutralize multivalent ions and create space between particles
4. Phosphonates: an example of a complexing agent
One of the most widely used complexing agents in ceramics is phosphonates. Their ability to eliminate multivalent charges enables efficient cation exchange, helping to space particles out and reduce system viscosity.
Phosphonates (or phosphonic acids) are a class of compounds that include at least one phosphonic group, featuring a direct phosphorus-to-carbon bond. This structure makes them chemically stable and resistant to reactions like hydrolysis and oxidation—ideal for use in demanding industrial environments. Applications of phosphonates (most important fields):
Water treatment: Prevent scale buildup and corrosion in piping and heating/cooling systems by binding to metal ions, blocking limescale formation
Detergents and cleaners: Used as sequestrants to trap calcium and magnesium, boosting detergent performance in hard water
Agriculture: Some phosphonates serve as fertilizers or plant protection agents, helping plants resist stress and fungal diseases
Polymer industry: Improve stability in plastics or are used in polymer production
WHAT HAPPENS WHITOUT COMPLEXING AGENTS?
Just like in the game of Pac-Man, phosphonates have the ability to capture and then release multivalent cations like calcium or magnesium, which tend to form unstable complexes.
Their effectiveness is usually very high—even small amounts of phosphonates can get the job done.
If a complexing agent isn’t added during the ceramic production process, the consequences can vary depending on the specific application.
- In glazes: Calcium and magnesium from the glazes dissolve into the milling water, leading to an increase in the glaze’s viscosity. This happens because the particles in the mill become surrounded by positive charges, which promotes agglomeration—unless a complexing agent is used to prevent it.
- In clay bodies: Without agitation and without complexing agents, the paste can take on a flan-like consistency. This is due to electrostatic attraction between the particles, which increases viscosity and can even lead to the system gelling.
Complexing agents can be supplied in liquid or solid form. In either case, they help stabilize suspensions, reduce thixotropic effects, and save both time and energy during the milling phase.

