#32 Foam formation within ceramic glazes: how to deal with it?
- Introduction
- Foam: definition
- Foam: pre-conditions
- Foam: managing
1. Introduction
Among all problems that may arise along the glazing line, the excessive foaming of ceramic glaze is one of the most frequent and serious.
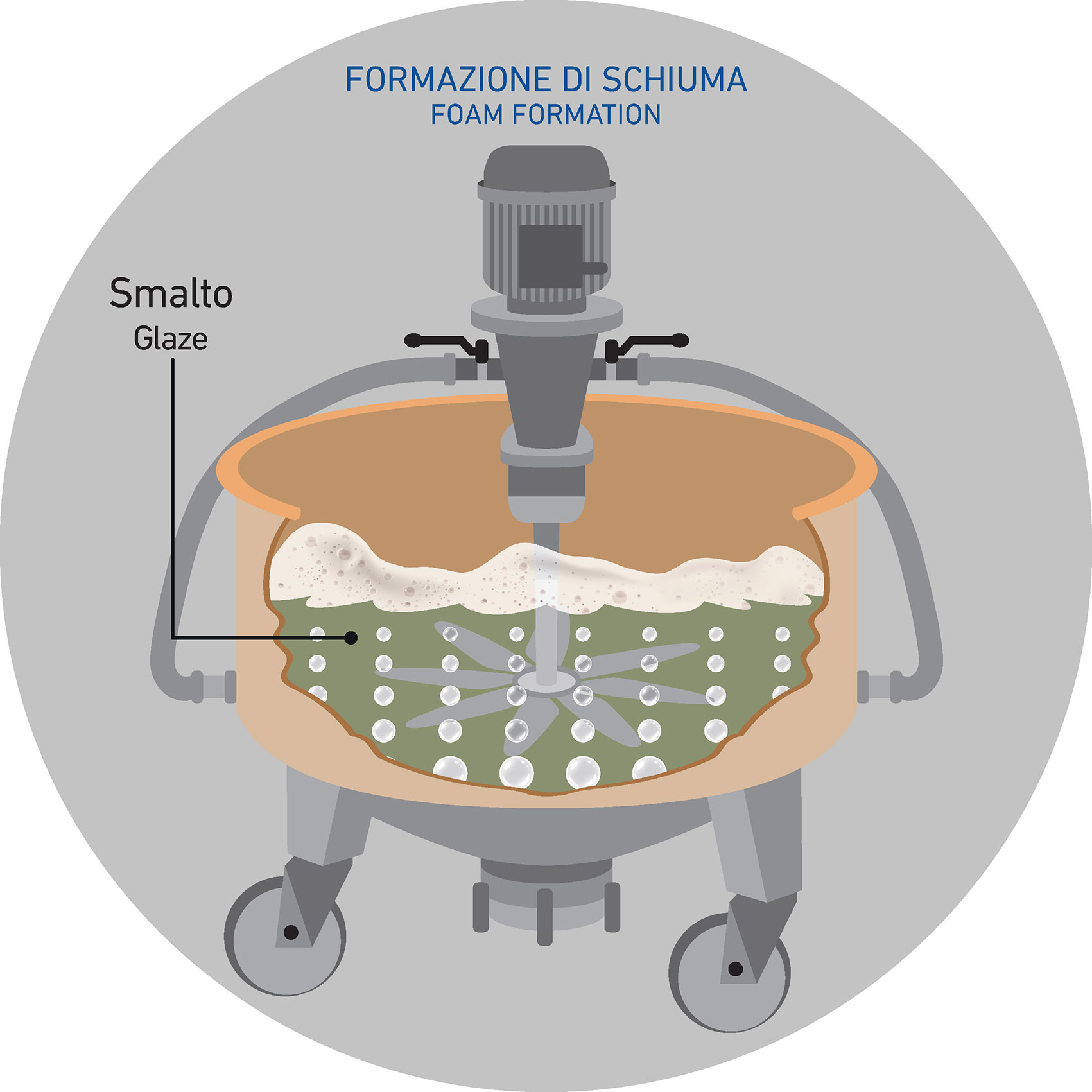
The phenomenon occurs and is visible on the glaze’s surface already within the stirring systems where the glaze is constantly under agitation to avoid sedimentation phenomena.
However, foam bubbles can sometimes be present not only during application but also at the end of the production process, that is after the firing cycle. This means that problems caused by foam may be also reflected on the finished product with the presence of superficial bubbles on the surface of the tile.
2. Foam: definition
From a chemical point of view, how can we define the foam? Here’s the simplest definition: dispersion (heterogeneous mixture of several phases) of a gas in a liquid, solid or gel medium.
The most classic example is the foam generated by the energetic movement of sea waves, where water (liquid phase) incorporates large quantities of air (gas phase) producing bubbles.
If we exclude ceramic production field, the presence of foam can also be a plus and therefore deliberately produced. In this case it can be generated by direct insufflation of a gas at high pressures or, for example, through the use of special foaming agents.
There are both liquid foams, such as those used by fire extinguishers, and solid foams, such as polyurethane foam or polystyrene. In general, foams have a messy structure marked by a variety of bubbles of different sizes.
3. Foam: pre-conditions
Several are the preconditions that can promote the formation of foam but – in ceramics – two are the most important:
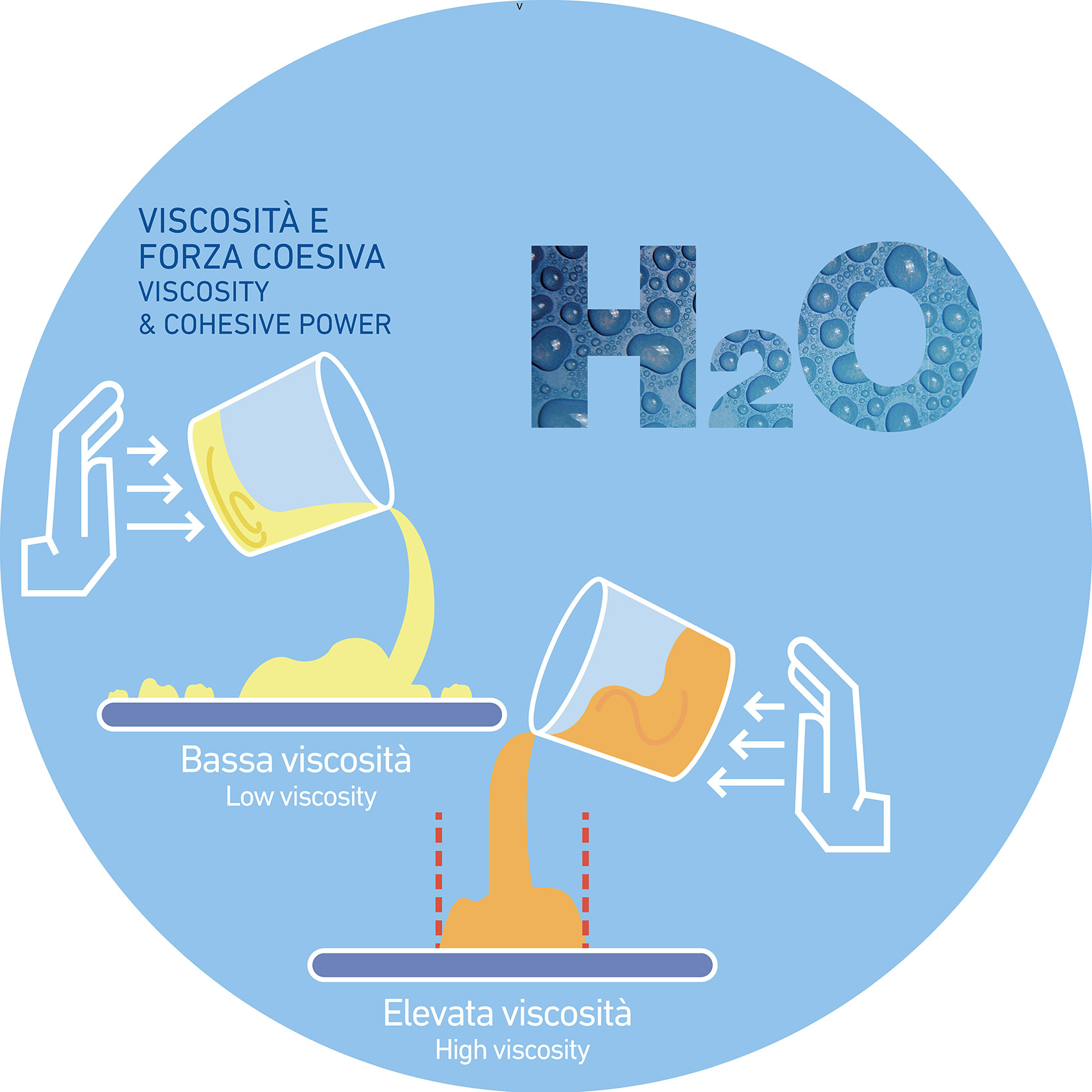
- Low viscosity of the glaze that makes the fluid more susceptible to the water movement within the stirring tank
- High water amount that can promote (or keep) a high surface tension within the suspension that in turn may hinder the degassing process, preventing the bubbles to explode
If these are the preconditions, what can act as a trigger? There’s a lot of different scenarios but the most frequent are:
- Presence of inorganic raw materials that contains surfactants deriving from previous processing of the producers of these raw materials
- Presence of chemicals marked by dispersant / wetting / binding action involved in the grinding process or added along the glazing line
Why?
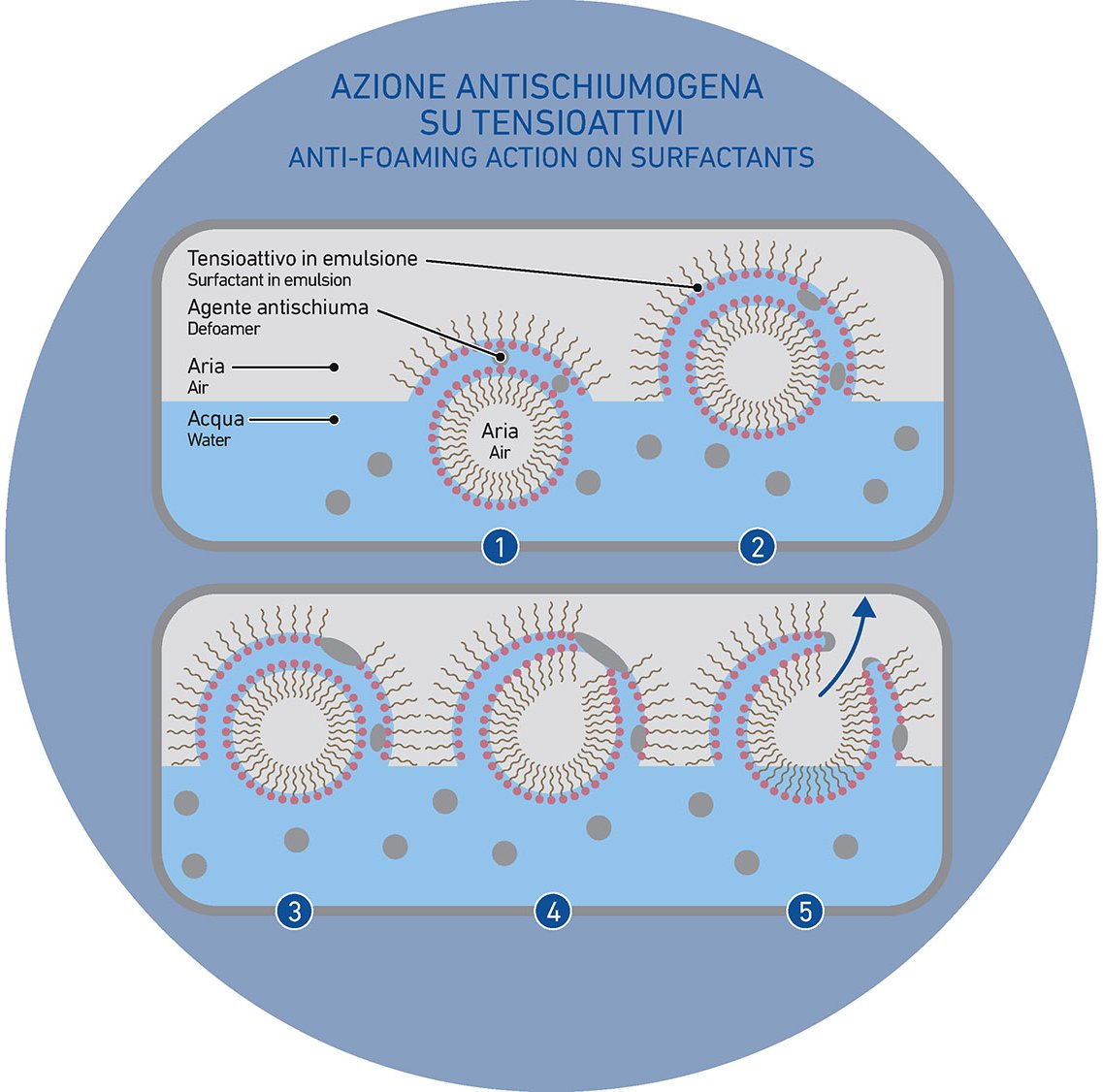
SURFACTANTS are high-performing soluble products that, for their very nature, are able to decrease the surface tension of the systems thanks to their molecular structure (consisting in a hydrophilic Head and a hydrophobic Tail). The decrease of the surface tension facilitates the production process and solves several problems but, in many cases, surfactants can be foamy and hard to control.
What is the process that leads to foam formation and what is the structure of a bubble?
Bubbles are spherical films consisting of water molecules that are held together by surfactants. In ceramics, when the glaze suspension is under stirring, the air circulates inside the glaze and foam bubbles arising to the surface.
In general, even in absence of surfactants, foam may appear due to the presence of molecules that can perform similarly to surfactants, Foam phenomena, in fact, are attributable not only to surfactants. This is the reason why it is important to be very careful during the formulation of chemicals inside the lab, trying to avoid potentially foaming products.
4. Foam: managing
When foam is little, it is better to avoid the use of other chemicals, to not compromise the rheological features of the glaze. Vice versa, when the problem is out of control, the use of antifoam is a must. In the most extreme cases, when the scenario is compromised, it could be necessary to rework on the glaze formulation.
What action must be taken to eliminate the problem?
There are many options but, in general, then most important and effective action involves the use of hydrophilic products able to interact with the molecules that have generated the foam. The interaction breaks the stability of the system, leading to bubbles burst. However, it is also possible to act by using water insoluble (and so hydrophobic) products. This is the case of defoamers that destabilize the foaming system affecting the surface tension and, therefore breaking the hydrophobic tails of the surfactant the holds the bubble, until it explodes.
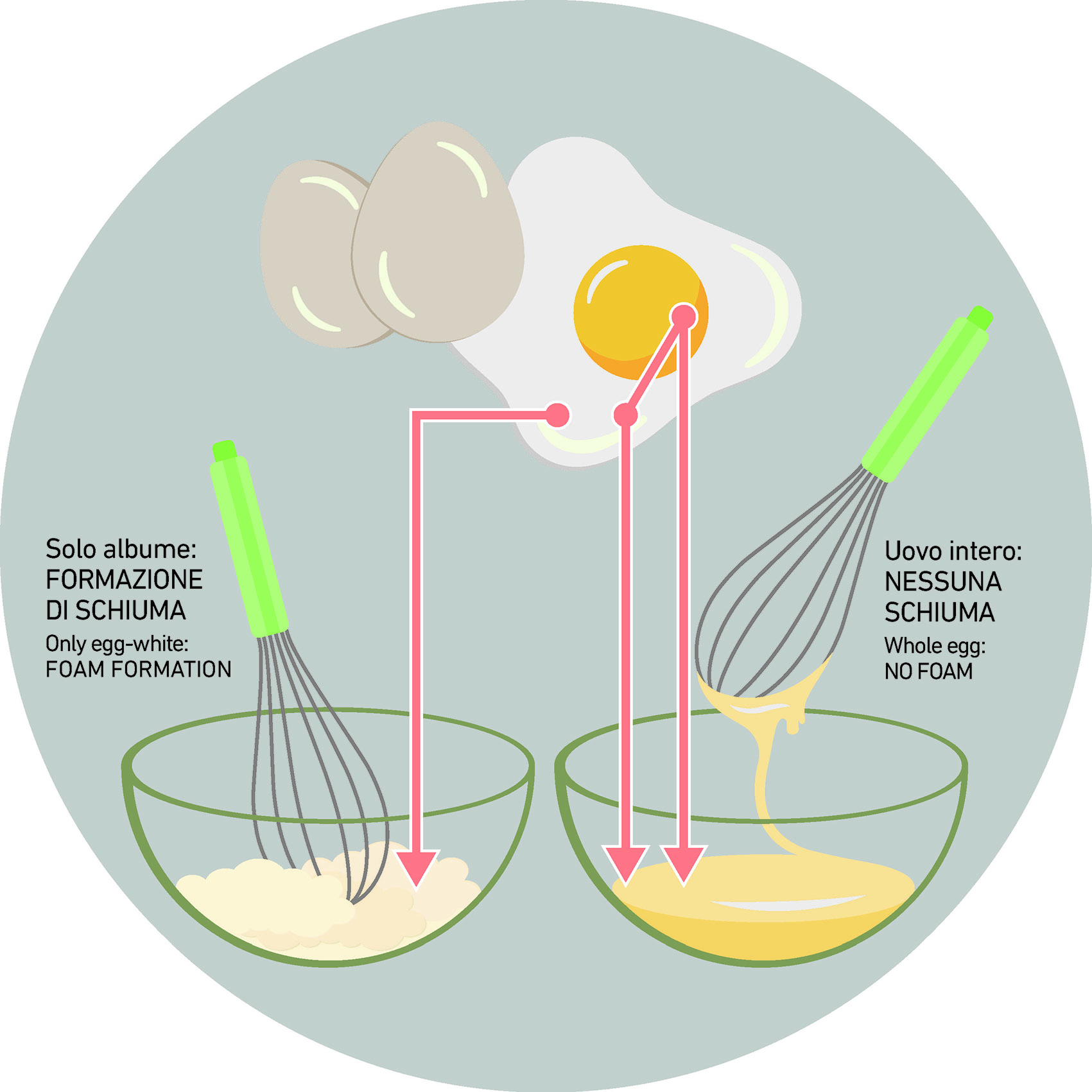
An example from the world of cooking.
Take an EGG and let’s start to mount it.
If we proceed by using only the egg white, we will notice the formation of foam. If we match the egg white together with the yolk (the red part) we will see the lack of foaming phenomena. Why? Because egg yolk contains very high percentages of fat (and in particular triglycerides) that act exactly as a defoamer, preventing the foam formation.